Researchers discover a mechanism allowing immune cells to regulate obesity
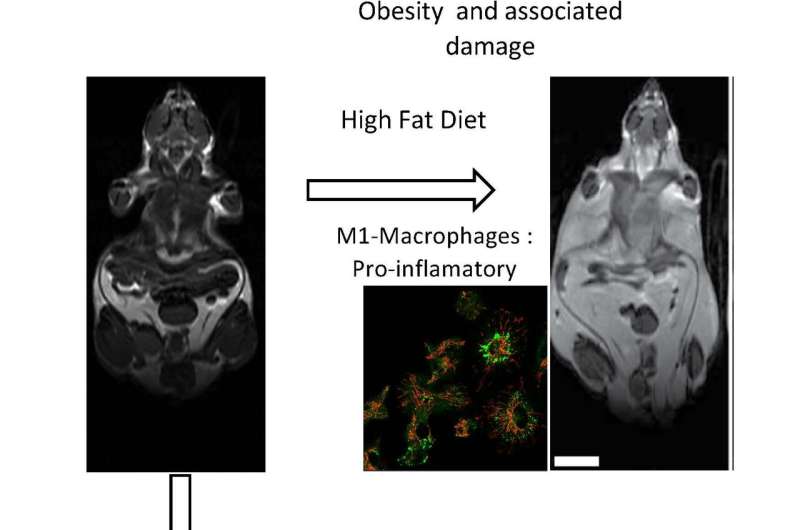
Macrophages are immune system cells. They are essential in the early response to infections, and they also have a key role in the proper functioning of our tissues and the regulation of obesity. Now, researchers at the Centro Nacional de Investigaciones Cardiovasculares (CNIC) have shown how this regulation unfolds in a paper published in Nature Metabolism, which could be useful to design new treatments for the obese and overweight, and for some associated pathologies, including fatty liver disease and type 2 diabetes.
The study was led by CNIC researchers directed by Dr. José Antonio Enríquez and Dr. David Sancho. It was completed in collaboration with the David Geffen School of Medicine and the Department of Medicine/Division of Cardiology of the University of California, Los Angeles (UCLA), in the US; the University of Eastern Finland and the Kuopio University Hospital (Finland); and the University of Salamanca and the Complutense University of Madrid. It explains how the activation of the mitochondrial metabolism of macrophages in response to oxidative stress due to excess nutrients contributes to fatty tissue inflammation and obesity.
"In recent decades, several studies have verified that fatty tissue macrophages facilitate an anti-inflammatory and reparative environment in normal conditions. This contributes to deactivating any processes altering the normal functioning of these tissues. These are known as anti-inflammatory or 'type M2' macrophages," Dr. Enríquez explains. In certain cases, he adds: "The M2 macrophages interpret that there are stress signals, normally arising in response to infection, and they foster inflammation as a defense mechanism."
These inflammation processes sourced to macrophages, says Dr. Enríquez, are responsible for the emergence of fatty tissue alterations, and "are the origin of obesity and the metabolic syndrome associated to cardiovascular disorders, fatty liver disease and type 2 diabetes." This means that, as a response to the excess nutrients created by a high-fat diet, "macrophages change their function and support inflammatory processes, forming 'type M1' proinflammatory macrophages."
Mitochondrial metabolism changes
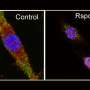
The research now published has analyzed how macrophage metabolic changes regulate this inflammatory process, which underlies obesity and the metabolic syndrome. The new findings, says Dr. Rebeca Acín-Pérez (currently at UCLA), "reveal how the detection by macrophages of oxidative danger signals—known as reactive oxygen species—leads to mitochondrial metabolism changes of these immune cells, needed to distinguish them from an M1 proinflammatory type." This oxidative stress, she clarifies, is found in morbidly obese patients, and it seems to be related to a high-fat diet, commonplace in the inadequate Western diet.
One of the conditions of this study, Dr. Sancho says, is that it proves that when this oxidative stress is reduced, "it ameliorates some of the harmful parameters associated with obesity."
In previous studies, CNIC scientists had found that the Fgr protein is decisive in regulating one of the complexes of the transport chain of mitochondrial electrons—the II complex—in response to this oxidative stress, and to benefit the generation of signals (cytokines and metabolites) fostering immune responses.
Salvador Iborra says that this study "proves that this same molecular mechanism regulates the conversion process of an anti-inflammatory macrophage (M2) governing the function of the tissue to a proinflammatory macrophage (M1), where lipid droplets accumulate. A balance between both types of M2/M1 macrophages is crucial for the proper functioning of the body."
Although inflammation is a normal body response and it is beneficial to face acute and transitory threats, it is very damaging when it becomes persistent or chronic, even in low-grade inflammation scenarios. The researchers explain that this happens in obesity and the metabolic syndrome, and it leads to increased cardiovascular mortality and diabetes.
The information contained in this new paper proves that, in the absence of the Fgr protein, the liver increases its ability to eliminate fat by generating ketone bodies (chemical compounds produced by ketogenesis, a process using body fats as an energy source), which are eliminated in the urine, and that this further enhances the alterations of obesity to the glucose metabolism (type 2 diabetes).
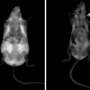
The results, found in mice, have been corroborated by human cohorts, where the authors found a stark correlation of Fgr and the negative consequences of obesity.
The researchers conclude that their data suggest the potential of using specific Fgr protein inhibitors to treat obese and/or metabolic syndrome patients. The goal would be reducing the associated inflammation, thereby improving the parameters associated with these illnesses, like fatty liver and type 2 diabetes, and contributing to raise patients' life expectancy and quality.
Obesity is a major health problem, and it is involved in the development of heart diseases, cerebrovascular accidents, cancer, fatty liver disease, metabolic syndromes, high blood pressure and some autoimmune diseases. A combination of an excess intake of nutrients, a lack of physical activity and genetic risk factors leads to an imbalance of energy demanded vs. energy consumed, and this is where obesity starts. In Spain alone, it is expected that in only a decade (by 2030), there will be 27 million obese and overweight adults (80% men and 55% women).
More information: Rebeca Acín-Pérez et al, Fgr kinase is required for proinflammatory macrophage activation during diet-induced obesity, Nature Metabolism (2020). DOI: 10.1038/s42255-020-00273-8