Team confirms existence of residual non-functioning beta cells in living individuals with longstanding type 1 diabetes
Scientists have found that a non-invasive imaging technique can detect residual, non-functioning beta cells in living individuals with longstanding type 1 diabetes—a breakthrough that brings us one step closer to the promise of beta cell regeneration—restoring insulin-producing cells lost in type 1 diabetes.
The new research is being presented at the Annual Meeting of the European Association for the Study of Diabetes (EASD), held online this year.
Until recently, it was widely believed that within months to a few years after type 1 diabetes diagnosis, a complete destruction of the insulin-producing beta cells occurred. However, emerging evidence suggests that most individuals with type 1 diabetes maintain a low level of residual beta cells long after type 1 diabetes onset. Nevertheless, scientists have struggled to confirm the existence of such small numbers of non-functioning residual beta cells in living people with long-duration type 1 diabetes using conventional methods.
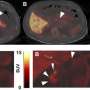
Single photon emission computed tomography (SPECT) is a non-invasive nuclear imaging technique that may provide such a method—involving measuring the pancreatic uptake of 111In-exendin (targeting the GLP-1 receptor abundantly expressed on beta cells).
"We hypothesized that type 1 diabetes patients may have considerable remaining beta cell mass, and therefore should have detectable tracer (111In-exendin) uptake in the pancreas, even without, or only low, insulin production", explains Professor Martin Gotthardt from Radboud University Medical Center in the Netherlands who co-led the research.
In the study, 10 adults (aged 21-54 years) with type 1 diabetes for an average of 11 years after diagnosis and 10 age- and sex-matched healthy controls were imaged with SPECT following injection of 111In-exendin to measure pancreatic tracer uptake.
To confirm the results obtained in living subjects, the research team then analysed organ donor pancreas ¬¬tissue from people with longstanding type 1 diabetes to assess GLP-1R expression, as well as presence of insulin and glucagon—another hormone which is often elevated in people with type 1 diabetes, and also contributes to poor glucose control.
Results showed that 6 out of 10 individuals living with type 1 diabetes had measurable pancreatic uptake of 111In-exendin. Strikingly, in half (5/10) of the type 1 diabetes patients tested, pancreatic tracer uptake was similar to the lower range of uptake levels in healthy controls, indicating that even after several years of type 1 diabetes, the function of these remaining beta cells could potentially be restored if adequate treatments were available.
Importantly, the findings were corroborated by histological analysis of pancreas samples from organ donors, that demonstrated the presence of a limited number of islets with residual insulin-positive beta cells, some of which were GLP-1R positive, even in those individuals with undetectable C-peptide (a marker for insulin production).
"The detected radiotracer uptake could indicate the presence of residual dysfunctional beta cells or, alternatively, GLP-1R expression on other endocrine cell types that may transform into a beta cell-like type", says Dr. Marti Boss from Radboud University Medical Center in the Netherlands who co-led the research.
According to Professor Gotthardt, "The presence of a residual pool of dysfunctional beta cells has important implications for treatment of type 1 diabetes, since these cells could help people maintain some ability to make their own insulin. These results are hugely encouraging, but we need to do more studies."
The authors point out that further investigations are needed to confirm these results and to better characterise the GLP-1R expressing cells.