New developments for the treatment of muscle spasticity after stroke and nervous system defects

Chronic muscle spasticity after nervous system defects like stroke, traumatic brain and spinal cord injury, multiple sclerosis and painful low back pain affect more than 10% of the population, with a socioeconomic cost of about 500 billion USD. Currently, there is no adequate remedy to help these suffering people, which generates an immense medical need for a new generation antispastic drugs.
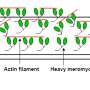
András Málnási-Csizmadia, co-founder of Motorpharma Ltd. and professor at Eötvös Loránd University in Hungary leads the development of a first-in-class drug candidate co-sponsored by Printnet Ltd. MPH-220 directly targets and inhibits the effector protein of muscle contraction, potentially by taking one pill per day. By contrast, current treatments have low efficacy and cause a wide range of side effects because they act indirectly, through the nervous system.
"We receive desperate emails from stroke survivors, who suffer from the excruciating symptoms of spasticity, asking if they could participate in our research. We work hard to accelerate the development of MPH-220 to alleviate these people's chronic spasticity," said Prof. Málnási-Csizmadia.
The mechanism of action of MPH-220 and preclinical studies are recently published in Cell. Dr. Máté Gyimesi, CSO of Motorpharma Ltd. highlighted: "The scientific challenge was to develop a chemical compound which discriminates between skeletal and cardiac muscle myosins, the motor proteins of these contractile systems. This feature of MPH-220 makes it highly specific and safe."
Prof. James Spudich, co-founder of Cytokinetics, MyoKardia and Kainomyx, all companies developing drugs targeting cytoskeletal components, is also very excited about MPH-220 as a possible next generation muscle relaxant. "Cytokinetics and MyoKardia have shown that cardiac myosin is highly druggable, and both companies have potential drugs acting on cardiac myosin in late phase clinical trials. Skeletal myosin effectors, however, have not been reported. Motorpharma Ltd. has now developed a specific inhibitor of skeletal myosin, MPH-220, a drug candidate that may reduce the everyday painful spasticity for about 10% of the population that suffers from low back pain and neurological injury related diseases," said Professor Spudich, former chair of Stanford medical school's Biochemistry department, a Lasker awardee.
Drug development specifically targeting myosins is becoming a distinguished area, as indicated by last week's acquisition of MyoKardia by Bristol-Myers Squibb Co. for 13.1 billion dollars in an all-cash deal, in the hope of marketing their experimental heart drug targeting cardiac myosin. This business activity shows the demand for start-up biotech companies such as Myokardia or Motorpharma.
"Motorpharma focusing on muscle research is ready to offer an antispastic drug candidate, MPH-220 with a new mechanism of action. The treatment of spasticity is an unmet need causing huge burden in diseases like poststroke conditions or chronic low back pain" said Prof. Istvan Bitter, former head of CNS in a regional hub of Eli Lilly Co.
More information: Máté Gyimesi et al. Single Residue Variation in Skeletal Muscle Myosin Enables Direct and Selective Drug Targeting for Spasticity and Muscle Stiffness, Cell (2020). DOI: 10.1016/j.cell.2020.08.050