Emergent: FDA letting troubled COVID vaccine factory restart

The U.S. Food and Drug Administration is allowing the problem-plagued factory of contract manufacturer Emergent BioSolutions to resume production of COVID-19 vaccine, the company said Thursday.
The Baltimore factory was shut down by the FDA in mid-April due to contamination problems that forced the company to trash the equivalent of tens of millions of doses of vaccine it was making under contract for Johnson & Johnson. The bulk vaccine was contaminated with an ingredient for AstraZeneca's COVID-19 vaccine, which was being made in the same factory.
The process of restarting operations began Thursday, but it will take time for the factory to ramp up and reach the production level of before the shutdown, according to Emergent spokesman Matt Hartwig.
Since April, FDA inspectors have combed the factory and reviewed security camera footage to identify lapses in handling material inside the factory and worked with Gaithersburg, Maryland-based Emergent to resolve a host of problems, including unsanitary conditions and poorly trained workers.
The agency also has been reviewing data on months' worth of vaccine substance made in the factory and has allowed several large batches of vaccine doses made from it to be released for distribution.
Shares of Emergent rose $1.31, or 2%, to $66.47 in afternoon trading Thursday.
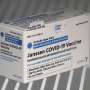
Emergent is one of several J&J contractors that produce its one-shot vaccine in bulk. The concentrated vaccine then is shipped to other factories for final steps, including diluting them to the correct strength, putting them in vials and packaging them up.
The lapses at the Bayview factory have hampered J&J's efforts to be a major player in vaccinating people, particularly in remote areas and poor countries, given that it's the only drugmaker with an authorized vaccine that only requires one dose and standard refrigeration. It's also cheaper than some other vaccines.
The productions problems have forced J&J to import millions of doses from its factory in the Netherlands to the U.S. and to miss supply commitments.
Emergent Chief Executive Robert Kramer said the company had fallen short of the public's expectations.
Emergent's factory had a history of FDA citations for problems including mold, dirty walls and floors, poorly trained employees and an inadequate strategy to prevent contamination, yet it was given a huge role in COVID-19 vaccine production by the Trump administration. Emergent was handed a lucrative contract to make many millions of COVID-19 vaccines for both J&J and AstraZeneca at the Bayview factory.
The Biden administration has been working to find a different American manufacturing partner for British drugmaker AstraZeneca, whose COVID-19 vaccine isn't authorized for distribution in the U.S.
© 2021 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.