Meniscus implant superior to non-surgical care for patients with pain after surgery
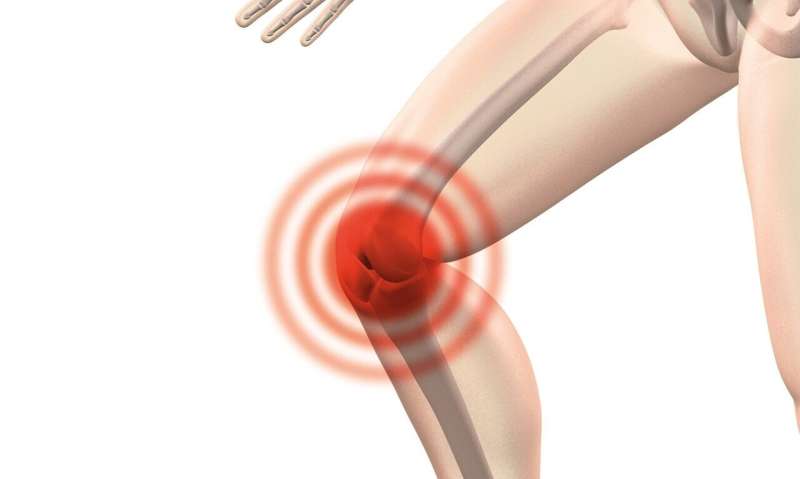
Results from two prospective, concurrent, clinical trials demonstrate that a synthetic meniscus implant provides superior relief from post meniscus surgery knee pain when compared to treatment with non-surgical care alone.
The research was presented today at the American Orthopedic Medicine Society- Arthroscopy Association of North America Combined 2021 Annual Meeting by Wayne Gersoff, MD, from Advanced Orthopedic & Sports Medicine Specialists, Parker, Colo.
Arthroscopic partial meniscectomy is the most common surgical treatment for symptomatic, irreparable meniscal tears that do not improve with non-surgical care. For many patients, arthroscopic partial meniscectomy is associated with improvements in knee pain and function, but a subset of patients reports persistent or recurrent knee pain 1-2 years after surgery. Current treatment options for post- arthroscopic partial meniscectomy knee pain are limited, especially for patients considered too young for knee replacement.
To broaden treatment options for these patients, two FDA-regulated clinical trials were conducted in the United States and pooled for analysis, to compare a synthetic medial meniscus implant to non-surgical care in treating persistent or recurrent knee pain following previous partial meniscectomy. The polymeric medial meniscus implant mimics the biomechanical function of the natural medial meniscus and is intended to provide relief from pain and improved function in subjects with knee pain following APM.
Researchers from three centers enrolled 242 patients (176 investigational, 66 control) were treated in two prospective, concurrent clinical trials in the U.S.: a randomized controlled superiority trial comparing the medial meniscus implant to non-surgical care (control) and a single-arm, implant-only trial.
Eligible subjects had persistent knee pain and one or more previous partial meniscectomies at least 6 months before trial entry. Treated subjects returned for follow-up visits at one month, six months, one year, and two years. Patient-reported knee pain, function, and quality of life were assessed using the Knee Injury and Osteoarthritis Outcome Score (KOOS).
Patients enrolled in the implant arm of the trial reported a 63.4 percent higher KOOS score than the control group, which reported a KOOS score of 27.9 percent. Additionally, more patients in the control group opted to stop treatment (17.3 percent) compared to the implant group (10.5 percent). This difference was not statistically significant.
"Two-year results from two prospective, concurrent, US clinical trials demonstrate that the synthetic meniscus implant provides superior relief from post-APM knee pain when compared to treatment with non-surgical care alone," reported Dr. Gersoff. "Overall, the implant group experienced superior improvements and outcomes at 2 years compared to subjects receiving non-surgical care alone."