Discovery points to new ways to kill aggressive cancer cells
Vanderbilt faculty and researchers are looking for the "Achilles' heel" of the cancer cells that survive initial chemotherapy. Michael King, chair of the Department of Biomedical Engineering, and Joshua D. Greenlee, graduate research fellow in biomedical engineering, are investigating whether a naturally occurring protein made in immune cells, called TRAIL, is effective in killing colon cancer cells.
This knowledge may enable the development of new therapies that make aggressive cancer cells easier to kill off before they spread to other parts of the body.
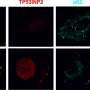
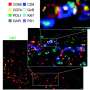
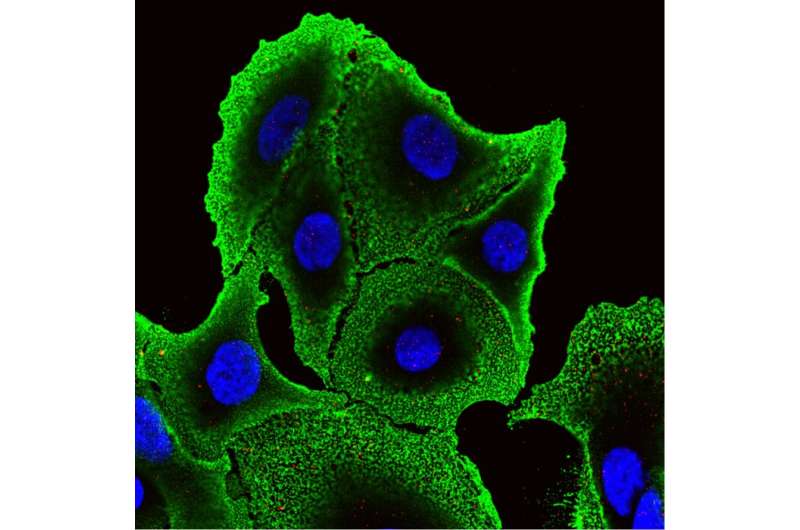
The researchers found that TRAIL is more effective in killing these drug resistant colon cancer cells. "We found that these cells had higher quantities of a protein called death receptor 4 on the cell surface," Greenlee said. "This receptor does just what the name suggests, causes death of the cell upon binding to TRAIL. We also found that these death receptors were more likely to cluster together on the cell membrane in what are known as 'lipid rafts.' When these receptors are found within these rafts, TRAIL is even more effective at killing cancer cells."
Using this knowledge, King, Greenlee and their collaborators designed TRAIL-coated nanoparticles that effectively kill 57 percent of cancer cells in the blood of patients with advanced chemo-resistant colon cancer. In some patient blood samples, the TRAIL-coated nanoparticles destroyed all detectable cancer cells, King said. Higher levels of lipid raft death receptor 4 in these patients also coincided with increased treatment efficacy of TRAIL-coated nanoparticles.
When colorectal cancer spreads to other organs in the body, patients require chemotherapy. While chemotherapy is typically successful at killing cancer cells in early treatment, stronger surviving cells develop resistance to the treatment and can form new and more lethal tumors.
"These results are particularly encouraging for colorectal cancer patients that have failed chemotherapy," said King, who is J. Lawrence Wilson Professor of Engineering. "Patients with metastatic disease that have failed chemotherapy are often left with few treatment options. The use of TRAIL as a nontoxic nanoparticle becomes particularly appealing among these patients."
More research will be directed toward drug discovery efforts to change lipid raft compositions in cancer cells to influence their interactions with death receptors and increase the therapeutic benefits of TRAIL, King said. The long-term goal is to take this research from the bench to the bedside, starting by testing TRAIL-coated nanoparticles in pre-clinical models.
The article, "Oxaliplatin resistance in colorectal cancer enhances TRAIL sensitivity via DR4 upregulation and lipid raft localization" was published in the journal eLife on August 3.
Greenlee, King and colleagues recently published a closely related review article, "Rafting Down the Metastatic Cascade: The Role of Lipid Rafts in Cancer Metastasis, Cell Death, and Clinical Outcomes" in the journal Cancer Research. The article covers the roles of lipid rafts in cancer metastasis.
More information: Joshua D. Greenlee et al, Rafting Down the Metastatic Cascade: The Role of Lipid Rafts in Cancer Metastasis, Cell Death, and Clinical Outcomes, Cancer Research (2020). DOI: 10.1158/0008-5472.CAN-20-2199