When less is more: A modified chemotherapy regimen for a rare type of lymphoma
Burkitt lymphoma (BL) is a highly aggressive form of non-Hodgkin lymphoma (NHL) that comes in three different variants: endemic, immunodeficiency-associated, and sporadic. Of these variants, sporadic BL is most commonly found outside malaria-endemic countries and typically seen in young adults.
Over the past two decades, progress in chemotherapy regimens for BL combining multiple drugs have led to increased survival rates. In particular, the CODOX-M and IVAC regimens have proved effective for the treatment of sporadic BL. Unfortunately, these standard regimens are short-term and intensive, which frequently causes high toxicity and severe, sometimes life-threatening complications. Moreover, these regimens were evaluated through clinical trials conducted in Europe and the United States; in other words, they may not be ideal for Asian populations.
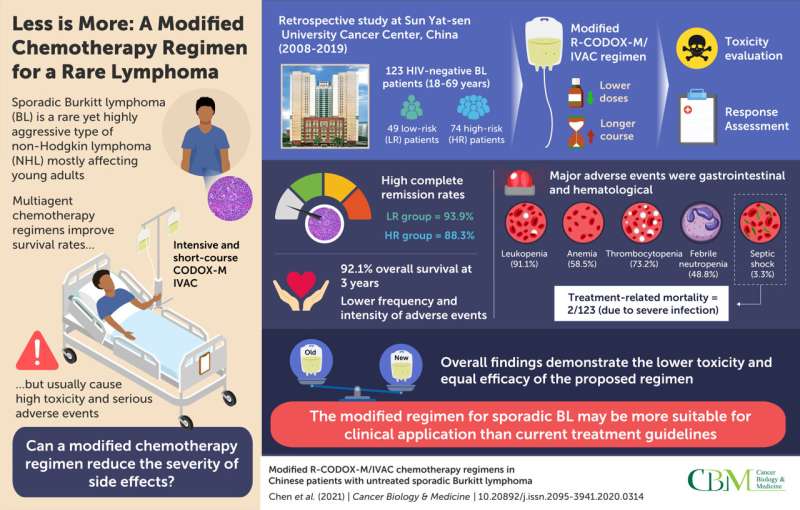
In a study recently published in Cancer Biology & Medicine, scientists from Sun Yat-sen University Cancer Center (SYSUCC), China, sought a solution to these problems. Based on their experience and the results of previous studies, they hypothesized that a CODOX-M/IVAC regimen with lower dosage but a longer exposure time would be just as effective as the recommended treatment but with a reduced resulting toxicity. Their proposed approach also included rituximab, a drug used to treat lymphomas, and is therefore referred to as the modified R-CODOX-M/IVAC regimen.
To test their hypothesis, the scientists conducted a retrospective study in which 123 HIV-negative patients with untreated sporadic BL were enrolled at SYSUCC between 2008 and 2019. The patients were divided into low-risk and high-risk groups; low-risk patients received four to six cycles of R-CODOX-M, whereas high-risk patients received six to eight alternating cycles of R-CODOX-M and IVAC. The researchers registered all adverse events likely caused by the chemotherapy treatment and calculated their relative frequencies.
Overall, the results for the modified regimen were promising. Not only was the overall survival rate after five years relatively high, but also the frequency and severity of adverse events were lower than those reported in previous studies for standard regimens. Serious mucositis, one the most concerning and frequent side effects of standard regimens, did not occur at all in this study. Moreover, sepsis cases were only four, and treatment-related mortality among the high-risk patients was two out of 74 patients.
"Our findings add to the growing body of non-randomized data demonstrating the efficacy and safety profile of modified R-CODOX-M/IVAC regimens," says Dr. Tongyu Lin of SYSUCC, who led the study, "Moreover, ours was the first report involving such a modified regimen in a cohort of adult Chinese BL patients."
The research team hopes that prospective trials will be conducted in the future using the proposed treatment strategy. "We believe that the modified R-CODOX-M/IVAC regimen may be more suitable for clinical application than the current guidelines and therefore deserves further evaluation," concludes Dr. Lin.
More information: Meiting Chen et al, Modified R-CODOX-M/IVAC chemotherapy regimens in Chinese patients with untreated sporadic Burkitt lymphoma, Cancer Biology and Medicine (2021). DOI: 10.20892/j.issn.2095-3941.2020.0314