Researchers identify key molecule that may lead to new treatments for COVID
Many treatments for COVID-19 focus on the spike protein that the virus uses to bind to human cells. While those treatments work well on the original variant, they may not be as effective on future ones. The Omicron variant, for example, has several spike mutations.
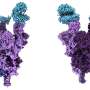
Pritzker School of Molecular Engineering Prof. Juan de Pablo and his group have used advanced computational simulations to examine another protein that's crucial to the virus's replication and remains relatively consistent across different coronaviruses. This protein, called Nsp13, belongs to a class of enzymes known as helicases, which play a role in how the virus replicates.
Through this work, the scientists have also uncovered three different compounds that can bind to Nsp13 and inhibit virus replication. Given the consistency of helicase sequences across coronavirus variants, these inhibitors could serve as a valuable starting point for designing drugs that target helicases in order to treat COVID-19.
"We currently only have one treatment for COVID-19, and as the virus mutates, we absolutely need to be targeting different building blocks besides the spike protein," de Pablo said. "Our work has revealed how small molecules are able to modulate the behavior of an attractive target in virus replication, and has shown that existing molecular scaffolds are promising candidates for COVID treatment."
The results were published in the journal Science Advances.
Disrupting a communication network
For the past two years, de Pablo and his group have used advanced computational simulations to study proteins that allow the virus that causes COVID-19 to replicate or infect cells. The simulations, which require months of extremely demanding computations with powerful algorithms, ultimately reveal how the virus works at the molecular level.
In this project, the collaborators examined the protein Nsp13, which unwinds double-stranded DNA into two single strands—a critical step in replication. Previously, researchers knew that Nsp13 performed this unwinding, but did not have a good understanding of the complicated dynamics of the process. The simulations revealed how multiple domains within the protein communicate with each other and act in concert to exert the right forces for the unwinding.
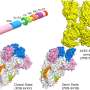
They also found that the moment an outside molecule binds to certain sites of the protein, it disrupts this communication network. That means the protein can no longer unwind the DNA efficiently and it becomes more difficult for the virus to replicate.
Several compounds had already been reported as Nsp13 inhibitors, but the researchers selected three compounds to test within their simulations: bananin, SSYA10-001, and chromone-4c.
The researchers found that all three appeared to disrupt the Nsp13 protein effectively by binding to certain sites and disrupting the protein's network. Now, de Pablo and his collaborators are working with experimentalists to test their results in the lab.
A series of candidates to treat COVID-19
Previously, the group used computational analysis to reveal how the drug Ebselen binds to the virus's main protease, or MPro. In a different study, they also revealed how the antiviral drug remdesivir binds to and interferes with the virus. They also showed how the compound luteolin inhibits the virus's ability to replicate.
The researchers have even used the information from their simulations to design a new drug to treat COVID-19, which they hope to publish within the next few months.
"We continue to look at drugs that affect different parts of the virus, different proteins, then use experimental data to confirm their efficacy," de Pablo said. "We now have a series of candidates, and our newly designed drugs could be game changers for treating COVID-19 and novel coronaviruses in the future."
Other authors on the paper include Gustavo R. Perez-Lemus, Cintia A. Menéndez, Walter Alvarado and Fabian Byléhn.
More information: Gustavo R. Perez-Lemus et al, Toward wide-spectrum antivirals against coronaviruses: Molecular characterization of SARS-CoV-2 NSP13 helicase inhibitors, Science Advances (2022). DOI: 10.1126/sciadv.abj4526