Arterial thromboses: An innovative treatment approach
Scientists have shown in an animal model that the CXCL12 messenger could be suitable as a target structure for the treatment of blood-clotting disorders.
Thromboses—the medical term for blood clots—occur in veins or arteries. Arterial thromboses can lead to heart attacks or strokes. Blood platelets (thrombocytes) play a key role in the disease process. Doctors prescribe drugs to prevent these cells from aggregating.
"Although current therapies are efficient, they have the drawbacks that they can cause hemorrhaging or that thromboses can happen despite them," says PD Dr. Philipp von Hundelshausen from LMU's Institute for Cardiovascular Prevention and from the German Center for Cardiovascular Research (DZHK). "To develop new forms of therapy, it's important to understand pathomechanisms at the molecular level."
In the journal Blood, von Hundelshausen's team reports on a new signaling pathway of blood clotting, which hitherto it has not been possible to influence by means of drugs. At the same time, the researchers demonstrate how this pathway can be experimentally inhibited in order to reduce the risk of thrombosis. Co-author of the study is Professor Christian Weber, Director of the Institute for Cardiovascular Prevention.
A new blood clotting mechanism
"We put together various known pieces of the arterial thrombosis puzzle and developed a new hypothesis about the molecule CXCL12," reports von Hundelshausen. It was known that CXCL12 can activate cellular messengers of hemostasis, platelets, in vitro, which leads to blood clotting. Researchers also knew that this happens with CXCR4 as the binding site. Found on all platelets, this receptor is usually responsible for chemotaxis—the directed movement of cells along concentration gradients of certain chemicals. Equally, it is known that platelets produce CXCL12.
In earlier experiments, the researchers had discovered a molecule that binds to CXCL12 and inhibits this messenger. Based on the blueprint, they manufactured a synthetic version of the inhibitor. But it was still unclear whether CXCL12 was involved at all in arterial thromboses. The experiments merely showed platelet activation. And the origin of CXCL12 was also mysterious.
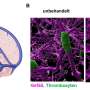
Using animal experiments, the LMU researchers were able to demonstrate that CXCL12 does in fact—as suspected—play a role in the genesis of thromboses. The experiments involved inflicting targeted chemical injuries on the carotid artery of mice.
Using gene technology, the scientists created a mouse model in which CXCL12 is produced everywhere it is normally produced, but not by blood platelets. Their results showed that thromboses formed later than in wild-type mice—and dissolved again faster. In genetically modified mice, blood vessels occluded less frequently.
As their next step, von Hundelshausen's team simulated the situation by chemically inhibiting CXCL12 with the previously manufactured molecule. When the researchers injected mice with the substance before inducing thrombosis, much fewer thromboses occurred—and the clots dissolved faster than in untreated animals.
The question remained as to what exactly happens in the inhibition of CXCL12. Further analyses showed that Bruton's tyrosine kinase plays a key role. This enzyme is a candidate for the treatment of leukemia, but is also involved in blood clotting: If you inhibit Bruton's tyrosine kinase with known oncological drugs, CXCL12 also loses its effect.
All study results come from laboratory and animal experiments. Nonetheless, the researchers point to an interesting possible application: After a heart attack, patients are given acetylsalicylic acid plus clopidogrel to prevent new blood clots from forming. "However, both drugs lead to incomplete platelet inhibition," says von Hundelshausen. "In certain situations, such as additional platelet activation through collagen, the new inhibitor could offer additional benefits without increasing the risk of bleeding."
More information: Julian Leberzammer et al, Targeting platelet-derived CXCL12 impedes arterial thrombosis, Blood (2022). DOI: 10.1182/blood.2020010140