Could releasing 'handbrake' immune cells help supercharge immunity?
WEHI researchers have found a way to supercharge immune cells that could enable them to clear disease and infections faster.
T cells are immune cells that help our bodies fight disease by seeking and destroying unhealthy cells. Regulatory T cells, or Treg cells, put the brakes on T cells, preventing them from misbehaving and attacking the body's healthy tissues.
Researchers have now discovered a way to release the "handbrake" hold that Treg cells have over normal T cells and intentionally put them into overdrive. Lifting this restraint could supercharge the response of T cells and lead to better treatment options for cancers and infections, where patients would benefit from rapid clearance of the unhealthy cells.
The research, led by Dr. Charis Teh, Simon Preston, Associate Professor Daniel Gray and Professor Marc Pellegrini, is published in Science Immunology.
Boosted response
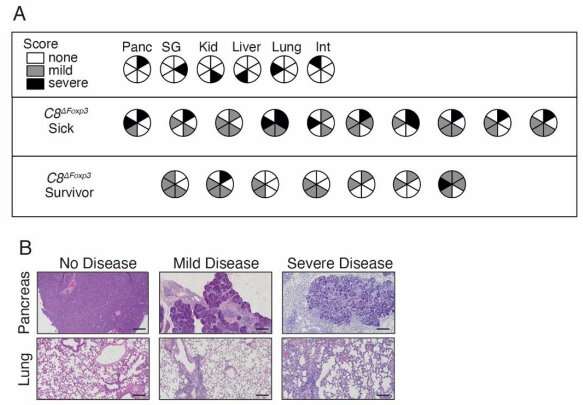
The team blocked the activity of an enzyme in Treg cells to assess its role in the immune response. They found the enzyme, known as caspase-8, controlled the survival of Treg cells in a manner that depended on the immune environment.
Restraining the enzyme in healthy settings enabled Treg cells to survive and accumulate. But when they blocked caspase-8 in Treg cells during an infection, this triggered a form of cell death called necroptosis.
Associate Professor Daniel Gray said inhibiting caspase-8 in Treg cells is a critical step that releases their restraint of T cells. "Releasing this 'handbrake' would greatly assist patients fighting certain debilitating conditions, such as chronic infections, as the T cells are then able to work overtime to drive the infection down or away. But there is risk when too many Treg cells die, as this can lead to autoimmune conditions like diabetes and multiple sclerosis.
"Knowing where the immune response can be supercharged to a level that will boost immunity, but not cause autoimmune disease, will be integral to advancing new immunotherapies in the future. Our research findings bring the scientific field closer to establishing this threshold."
A therapeutic window
By testing a clinically approved drug on human Treg cells, the researchers found they died more readily than normal T cells. This discovery identified a "therapeutic window" where Treg cells could be targeted for death while sparing normal T cells.
"If we were to target caspase-8 in Treg cells correctly, we could temporarily reduce their suppression on the immune system to enable better defense against certain infections and cancers," Dr. Teh said. "Our findings have revealed a new role for this enzyme that could be exploited to fine tune the balance of the immune response to pathogens, cancer and healthy, normal cells."
More information: Charis E. Teh et al, Caspase-8 has dual roles in regulatory T cell homeostasis balancing immunity to infection and collateral inflammatory damage, Science Immunology (2022). DOI: 10.1126/sciimmunol.abn8041