All high-grade noninvasive bladder cancers should be considered high-risk, study suggests
Noninvasive (stage Ta) bladder cancers with "high-grade" characteristics should be considered at high-risk of recurrence, concludes a study in The Journal of Urology.
All high-grade Ta cancers "behave similarly" in terms of response to initial treatment and risk of recurrence, regardless of other risk factors, according to the report Ashish M. Kamat, MD, of The University of Texas MD Anderson Cancer Center, and colleagues. That finding supports classifying all high-grade Ta tumors as high-risk, in contrast with current guidelines stating that these cancers should be further classified as high-risk or intermediate-risk.
Findings question recent change in risk assessment for Ta bladder cancer
The study included 251 patients with stage Ta bladder cancer treated between 2000 and 2018. Stage Ta cancers are noninvasive, meaning that they have not grown into the deeper layers (lamina propria or muscle) of the bladder wall. All patients received initial treatment with bacillus Calmette-Guérin (BCG): an immune therapy (non-chemotherapy) treatment that triggers the immune system to attack cancer cells.
Of the 251 Ta cancers, 202 were classified as high-grade: faster-growing tumors that are more likely to recur or progress after treatment. The remaining 49 cancers were classified as low-grade, with slower growth and a lower progression and recurrence rates.
Patients were then grouped into risk groups based on current European Association of Urology (EAU) guidelines, which use risk factors such as age, number of tumor foci, and tumor size. Using this system, about three-fourths of low-grade cancers were classified as intermediate-risk. Of the high-grade cancers, 50% were classified as high-risk and 45% as intermediate-risk. (The AUA has a similar set of guidelines, which use tumor foci and tumor size, but not age.) Dr. Kamat and colleagues analyzed the response to treatment and long-term outcomes based on tumor grade (high versus low) and patient risk (intermediate versus high).
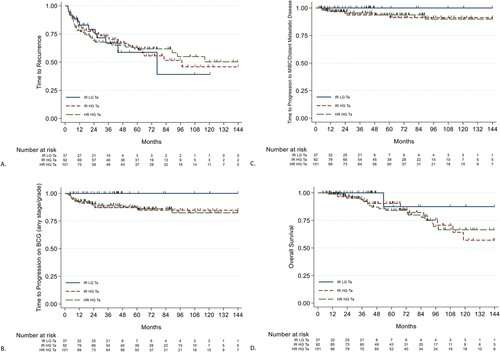
The results showed that patients with high-grade tumors had similar outcomes, regardless of risk grouping status. Based on the modern, accepted definition of BCG unresponsive disease, 100% of intermediate-risk, low-grade cancers responded to treatment with BCG. In contrast, 13% of high-grade, high-risk cancers and 14% of high-grade, intermediate-risk cancers were BCG unresponsive.
At a median follow-up of about 4 years, risk of cancer progression was also similar for all patients with high-grade Ta cancers regardless of the "risk grouping." Numerically, risk was actually higher in the intermediate-risk group: 6.5% for intermediate-risk and 5.9% for high-risk cancers. By comparison, none of the patients with low-grade Ta cancers had cancer progression.
Other outcomes were similar between groups, reflecting the generally good outcomes of BCG treatment for these noninvasive bladder cancers. Five-year survival rate without cancer spread (metastasis) was 100% for patients with low-grade, intermediate-risk Ta cancer and 94% for those with high-grade cancers, regardless of risk status.
About three-fourths of patients with bladder cancer have noninvasive disease when diagnosed. However, it is difficult to predict the risk of cancer recurrence and progression over time. Various risk classifications have been proposed, some of which classify Ta high-grade tumors as intermediate-risk, which might lead to some patients not receiving optimal therapy.
In this study, treatment response and cancer recurrence appear similar among patients with high-grade Ta cancers, regardless of whether they meet intermediate- or high-risk criteria. The clinical risk factors other than grade "are not sufficient to distinguish which patients are high-risk versus intermediate-risk," the researchers conclude.
Dr. Kamat comments, "These data support reverting back to the former EAU risk stratification model (2020 and prior), which recommended that all high-grade Ta lesions should be considered high-risk and receive appropriate treatment with intravesical BCG immunotherapy."
More information: Kelly K. Bree et al, All High-Grade Ta Tumors Should Be Classified as High Risk: Bacillus Calmette-Guérin Response in High-Grade Ta Tumors, Journal of Urology (2022). DOI: 10.1097/JU.0000000000002678