A promising new antibiotic for resistant gonorrhea
A new preclinical drug candidate developed by scientists at the Hackensack Meridian Center for Discovery and Innovation (CDI) shows great promise in eradicating the bacteria causing gonorrhea, according to a new publication.
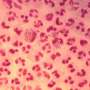
JSF-2659, developed to be administered orally, could be a game changer in treating Neisseria gonorrhoeae, which is on the World Health Organization's global list of "priority pathogens" for its resistance to existing drugs.
"This compound shows great promise, and is addressing an emerging health threat head-on," said David Perlin, Ph.D., chief scientific officer and senior vice president of the CDI. "We need more drugs, and better drugs. This molecular strategy could also very well have promise for pathogens beyond this one species of bacteria, as well."
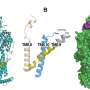
The parent compound JSF-2414 works simultaneously on two molecular targets, making it extra effective at inhibiting the bacteria's DNA replication, according to the paper in the journal Antimicrobial Agents and Chemotherapy.
The dual-targeting strategy means that more bacteria are wiped out—and the lack of survivors and very low probability of modifying two cellular targets during therapy means less chance for the rise of drug resistance, according to the various CDI models of the drug in action against N. gonorrhoeae.
Testing included 96 separate strains of the gonorrhea bacteria. But it also showed effectiveness in other Gram-positive germs like methicillin-resistant Staphylococcus aureus (MRSA), Staphylococcus epidermidis, Enterococcus faecium, Clostridium difficile, and other species.
The new drug candidate, if moved forward for clinical development in humans, would complement a list of drugs which have become less effective in recent decades. According to the paper, "there is now a high prevalence of N. gonorrhoeae strains that are resistant to common antimicrobial classes used for treatment including sulfonamides, penicillins, cephalosporins, tetracyclines, macrolides, and fluoroquinolones. Therapeutic failures with the extended-spectrum cephalosporins, such as cefixime and ceftriaxone, have created a major health crisis." In many developing countries, the "last-line" monotherapy drug is ceftriaxone.
Accordingly, gonorrhea is a "major public health concern" that infected more than 82 million new people worldwide last year, bringing with it its increasing resistance to such treatments, according to the World Health Organization. The WHO added multi-drug resistant gonorrhea to its "priority pathogens" list in 2017.
Research on the JSF-2659 compound was spearheaded by the CDI's Center of Excellence in Translational Research (CETR). Authors on the work included Perlin and members of his CDI lab, as well as veteran microbiologist Barry Kreiswirth, Ph.D., and members of his laboratory, and also colleagues include Joel Freundlich, Ph.D. from Rutgers University.
"The CDI's work continues to push the envelope in so many ways," said Ihor Sawczuk, M.D., FACS, Hackensack Meridian Health's president of Academics, Research and Innovation, and also associate dean of Clinical Integration and professor and chair emeritus of Urology at the Hackensack Meridian School of Medicine. "The world is better for their scientific contributions."
More information: Steven Park et al, A Novel Oral GyrB/ParE Dual Binding Inhibitor Effective against Multidrug-Resistant Neisseria gonorrhoeae and Other High-Threat Pathogens, Antimicrobial Agents and Chemotherapy (2022). DOI: 10.1128/aac.00414-22