Phase II study of Durvalumab plus concurrent radiotherapy in unresectable locally advanced NSCLC
Researchers from Japan today reported that the first phase II study to evaluate the efficacy and safety of durvalumab and concurrent curative radiation therapy for PD-L1-positive unresectable locally advanced non-small cell lung cancer without chemotherapy met its primary endpoint with tolerable adverse reactions. The research was reported today at the IASLC 2022 World Conference on Lung Cancer in Vienna.
Immunotherapy plays an important role in NSCLC and combination of radiotherapy and immunotherapy have been reported to have a synergistic effect. Durvalumab after concurrent chemoradiotherapy has been standard of care with unresectable locally advanced non-small cell lung cancer (stage III /postoperative recurrent NSCLC). But some patients are unable to complete concurrent curative radiation therapy and cannot receive durvalumab.
Dr. M. Tachihara from Kobe University Graduate School of Medicine, Kobe, Japan and colleagues developed the DOLPHIN study— the first Phase II study of immunotherapy combined with curative radiotherapy for unresectable locally advanced non-small cell lung cancer.
Dr. Tachihara and researchers at 12 research locations in Japan enrolled 35 adult patients with unresectable locally advanced non-small cell lung cancer with an ECOG performance status (PS) 0-1, PD-L1≥1% (SP263 clone). Patients received curative radiation therapy (60Gy) plus durvalumab 10 mg/kg every two weeks simultaneously, followed by maintenance with durvalumab for up to 12 months until disease progression (PD) or unacceptable toxicity.
Between September 2019 and November 2020, 35 patients were enrolled from 12 institutions in Japan. Of the 35 patients (median age, 72 years), 88.6% were male, 54.3% had ECOG PS 0, 96.1% had a history of smoking, 57.1% had non-squamous histology, and 25.7% were postoperative recurrence.
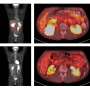
Thirty-four patients were evaluated for safety, and 33 patients for efficacy. The 12-month progression-free survival rate by ICR was 72.1% (90% CI, 59.1-85.1) after a median follow-up of 18.7 months. Confirmed overall response rate was 90.9% (95% CI, 75.7-98.1, ICR-assessed) with complete and partial response rates of 36.4% and 54.5%, respectively. Median progression-free survival was not reached by ICR-assessed, and 24.1 months (95% CI, 16.0-NR) by investigator-assessed. Thirteen patients (39.4%) discontinued durvalumab; six patients due to progression disease and seven due to adverse events (AE). Grade 3 /4 adverse events occurred in 47.1%; the most common adverse event of grade 3/4 was lung infection (11.8%) and pneumonitis (11.8%). Grade 5 adverse events of any cause occurred in 2 patients (5.9%), one with lung infection and one with broncho-esophageal fistula because of tumor progression during follow-up.
"This DOLPHIN study is the first report of immunotherapy combined with curative radiotherapy for unresectable LA-NSCLC. The primary endpoint of 12-months PFS rate was met and much higher than expected value. It suggests that this treatment strategy is promising with tolerable adverse effects and appropriate as a study treatment for phase III trials," said Dr. Tachihara.