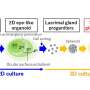
World's first mini organ transportation to a patient with ulcerative colitis
Tokyo Medical and Dental University (TMDU) research team announced on July 7 that it has succeeded in the world's first clinical transplantation of a "mini organ" (also called an organoid) into a patient with ulcerative colitis (UC). UC causes inflammation and ulcers (sores) in your digestive tract. It can be debilitating and can sometimes lead to life-threatening complications. UC belongs to a group of conditions called Inflammatory Bowel Disease (IBD). The number of patients is increasing in Japan, and worldwide is estimated to be about 5,000,000. The common treatment is to suppress inflammation with drugs, but in severe cases, the entire colon may be removed.
Dr. Mamoru Watanabe, vice president and distinguished professor of Tokyo Medical & Dental University said, "If our first-in-human research using organoids transplantation yields good results, we expect that the development of organoid medicine for intractable diseases of the digestive tract such as Crohn's disease will progress."
Dr. Ryuichi Okamoto, a professor of the Department of Gastroenterology and Hepatology, Graduate School of Medical and Dental Sciences said, "We embarked on the path of developing new methods for treating intractable diseases. This treatment should establish the efficacy and safety as soon as possible and deliver to the patients." If the team's effort is successful, the mucous membrane may regenerate and lead to a radical cure of UC.
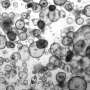
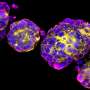
The clinical research started with collecting from the patient's vicinity of a healthy colonic mucosa and culturing them for about one month to form spherical organoids with a diameter of about 0.1 to 0.2 mm. On July 5, an organoid was transplanted into the colon of the same patient using a colonoscopy. The patient did well and was discharged July 6.
In previous experiments using mice models, the team confirmed that the cells were cultured in organoids and then transplanted, the mucous membranes regenerated in about a month and the clinical course improved, while the stem cells alone did not transplant because they were not able to culture in vitro.
In this clinical study, since the patient's own cells are used, there is an advantage that transplant rejection does not occur. In addition, since colonoscopy is used for collection and transplantation, there is no need for laparotomy, and the treatment can be performed in a minimally invasive method.
After this transplantation, medical examination will be conducted at the time after 4 weeks and 8 weeks. The patient will be monitored for up to a year to verify safety and efficacy. A further organoids transplantation is to be performed for up to eight patients.