A breakthrough in bacterial vaginosis treatment for women's health
The human microbiome has been a hot topic over the last decade, with research pointing to disrupted bacterial communities as culprits for a host of maladies including irritable bowel syndrome, eczema, and autoimmune diseases. Most studies have focused on the microbiome within the human gut, but there is growing recognition that another oft-ignored bacterial community deserves equal attention—that found in the vagina.
Vaginal microbiome disruptions cause bacterial vaginosis (BV), which afflicts nearly 30% of reproductive-aged women around the globe and costs an estimated $4.8 billion to treat annually. BV doubles the risk of many sexually transmitted infections including HIV and increases the risk of pre-term birth in pregnant women, which is the second-leading cause of death in newborns. BV is currently treated with antibiotics, but it often recurs and can lead to more severe complications including pelvic inflammatory disease and even infertility.
Just as probiotics are now being prescribed to treat gut issues, living biotherapeutics are being explored for the treatment of BV. However, it is difficult to conduct preclinical trials because the human vaginal microbiome is dramatically different from that of common animal models. Studies have found that Lactobacilli bacteria comprise more than 70% of the healthy human vaginal microbiome, but less than 1% of the vaginal microbiome in other mammals.
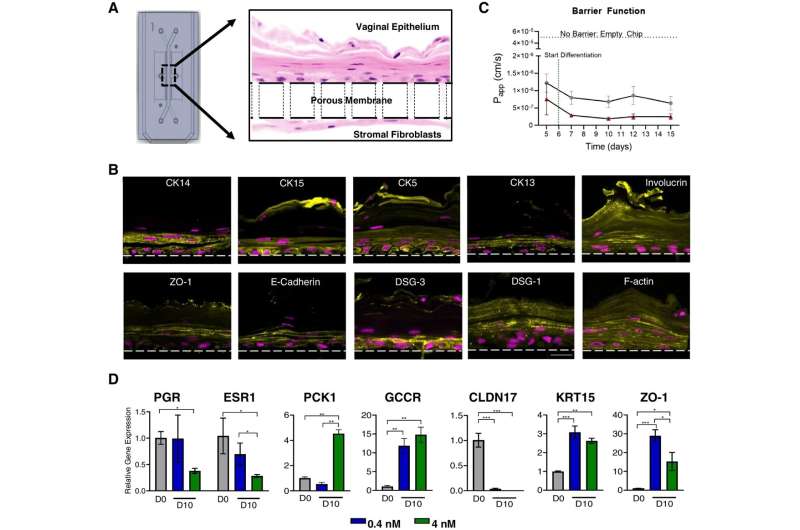
Researchers at the Wyss Institute at Harvard University have created a solution to that problem in the form of a new Organ Chip that replicates the human vaginal tissue microenvironment including its microbiome in vitro. Composed of human vaginal epithelium and underlying connective tissue cells, the Vagina Chip replicates many of the physiological features of the vagina and can be inoculated with different strains of bacteria to study their effects on the organ's health. The chip is described in a new paper published in Microbiome.
Modeling the vaginal microbiome
The Vagina Chip was developed with assistance from the Bill and Melinda Gates Foundation, which aimed to create a biotherapeutic treatment for BV and move it into human clinical trials to decrease infections of the reproductive tract, prenatal complications, and infant death rates. particularly in low-resource nations.
"A major stumbling block for that effort was that there were no good preclinical models that could be used to study which therapies can actually treat BV in human tissues. Our team's project was to create a human Vagina Chip to aid in the development and testing of new therapies for BV," said co-author Aakanksha Gulati, Ph.D., a Postdoctoral Researcher at the Wyss Institute.
Using the microfluidic Organ Chip platform developed at the Wyss Institute and subsequently licensed to Emulate, the team seeded the top channel of a polymer chip with human vaginal epithelial cells. They then added human uterine fibroblast cells to the opposite side of the permeable membrane separating the top and bottom channels. This 3D arrangement replicated the structure of the human vaginal wall.
After five days, the Vagina Chip had developed multiple distinct layers of differentiated cells that matched those found in human vaginal tissue. When the scientists introduced the female sex hormone estradiol (a form of estrogen) into the Vagina Chip, the chips' gene expression patterns changed in response, indicating that it was sensitive to hormones—another critical feature for replicating human reproductive organs in vitro.
Armed with a living model of the human vagina, the team then moved to study the vaginal microbiome. Recent research has shown that healthy human vaginal microbiomes typically contain multiple strains of Lactobacillus bacteria, so they worked with Dr. Jacques Ravel, Ph.D. and his team at University of Maryland School of Medicine, who had created three different consortia each containing several strains of L. crispatus. When they introduced these consortia into the Vagina Chip, all three successfully colonized the chips after three days. The consortia also began producing lactic acid, which helps to maintain the vagina's low pH and inhibits the growth of other microbes.
Beyond helping to maintain an acidic environment, the presence of the L. crispatus bacteria also affected the Vagina Chip's innate immune responses. Chips with bacterial consortia produced lower levels of several inflammation-causing cytokine molecules than chips without the bacteria, which is consistent with the current theory that these "good" microbes help keep inflammation in check in healthy human vaginas.
Bad bacterial tenants, on a chip
Having created a healthy Vagina Chip with optimal bacterial residents, the team then conducted a new experiment in which they inoculated chips with different species of bacteria that are associated with BV: Gardnerella vaginalis, Prevotella bivia, and Atopobium vaginae. A consortium of those three "bad" microbes caused the chips' pH to increase, damaging the vaginal epithelial cells and significantly increasing the production of multiple proinflammatory cytokines—all responses that were similar to what has been observed in human patients with BV.
"It was very striking that the different microbial species produced such opposite effects on the human vaginal cells, and we were able to observe and measure those effects quite easily using our Vagina Chip," said co-author Abidemi Junaid, Ph.D., a Research Scientist at the Wyss Institute. "The success of these studies demonstrate that this model can be used to test different combinations of microbes to help identify the best probiotic treatments for BV and other conditions."
The team is now using the Vagina Chip to test new and existing treatments for BV to identify effective therapies that can be advanced into clinical trials. They are also working on integrating immune cells into the chip to study how the vaginal microbiome might drive systemic immune system responses.
"There is growing recognition that taking care of women's health is critical for the health of all humans, but the creation of tools to study human female physiology is lagging," said senior author Don Ingber, M.D., Ph.D., who is the Wyss Institute's Founding Director.
"We're hopeful that this new preclinical model will drive the development of new treatments for BV as well as new insight into female reproductive health." Ingber is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children's Hospital and the Hansjörg Wyss Professor of Bioinspired Engineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.
More information: Gautam Mahajan et al, Vaginal microbiome-host interactions modeled in a human vagina-on-a-chip, Microbiome (2022). DOI: 10.1186/s40168-022-01400-1