New insights into medicines for preeclampsia
Burnet Institute researchers have led the development of the first global strategic guidelines—called target product profiles (TPPs)—for medicines to prevent and treat preeclampsia, a hypertensive disorder in pregnancy that's one of the leading causes of maternal deaths, stillbirth, and preterm birth globally.
Published in the journal PLOS Global Public Health, the TPPs will help to address a longstanding dearth of innovation, and research and development of new medicines for mothers, particularly for pregnancy-related conditions.
"Of the 300,000 global maternal deaths each year, 14 percent are due to hypertensive disorders, of which preeclampsia is the most common," Dr. McDougall, a Senior Research Officer in Burnet's Global Women's and Newborn's Health program, said.
"The vast majority of these deaths happen in low- and middle-income countries where women don't always have access to even the limited amount of medicines and quality of care available in high-income countries.
"So, in many cases, the only effective course of action is to deliver the baby, which also makes preeclampsia one of the leading causes of preterm birth."
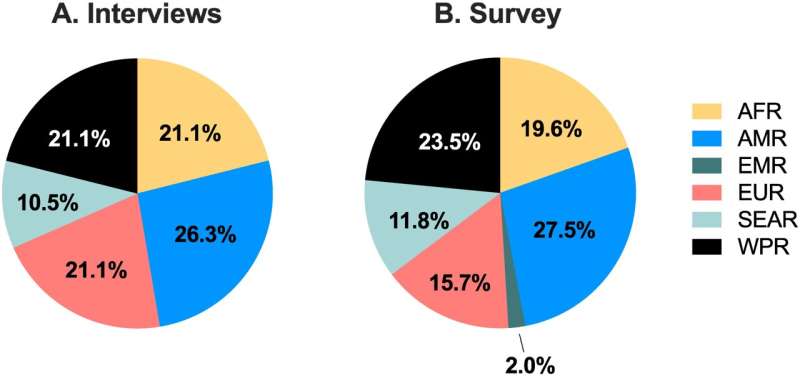
Developed through a process of consensus involving interviews, surveys, and feedback with global stakeholders, the new TPPs provide a detailed description of what new medicines for preeclampsia need to achieve to meet the needs of women and their health care professionals, particularly with a view to global implementation.
The TPPs would be used by pharmaceutical companies to inform the development of new drugs and products, and to assist funding agencies in their decision making.
"We found there's a high consensus globally on what these new medicines need to look like to be globally implemented," Dr. McDougall said.
"Using that information, we've identified the seven medicines with the highest priority, and eight that show medium potential, and this is where the field should be looking to in terms of where the next preeclampsia medicines might be coming from."
Dr. McDougall said women globally stand to benefit enormously from new medicines to prevent or treat preeclampsia.
"Women in Australia and the US or UK are less likely to die from preeclampsia, but their babies are still being born pre-term," she said.
"But we're trying to ensure that women in low- and middle-income countries at risk of preeclampsia don't miss out on new innovations."
More information: Annie Ra Mcdougall et al, Target product profiles for novel medicines to prevent and treat preeclampsia: An expert consensus, PLOS Global Public Health (2022). DOI: 10.1371/journal.pgph.0001260