Extending pre-quit duration of leading smoking cessation medication doesn't help smokers quit, study finds
Extending the duration that a leading smoking cessation medication is taken prior to stopping tobacco use does not significantly improve the likelihood of breaking the cigarette habit and staying smoke-free, according to the results of a study published in JAMA Network Open.
Varenicline is the best medication available for helping people to quit smoking. It has been sold since 2006 under the brand names Chantix and Champix, and recently became available in generic form.
But even with varenicline, most people return to smoking within six months of quitting, according to Larry Hawk, Ph.D., professor of psychology in the University at Buffalo College of Arts and Sciences and the study's first author.
"We are testing ways to improve quit rates," says Hawk.
Before the researchers conducted this clinical trial, there was preliminary evidence suggesting that improvement might materialize by asking adults to take varenicline for more than the typical one week before trying to quit.
But Hawk says the present data do not support that hypothesis.
"The simplest conclusion from this work is that we conducted a very strong, methodologically sophisticated and sound large-scale study, but abstinence rates at 3- and 6-month follow-up were not better for people taking varenicline for four weeks before trying to quit than for those with just one week of pre-quit medication."
The door, however, remains open on this line of research.
Though no one is certain why, women have a tougher time quitting smoking on average than men, according to Hawk, who says it would be worthwhile to conduct another study that focused exclusively on women.
"Our pilot study suggested the extended pre-quit approach worked better for women than men. Although that effect wasn't statistically significant in the current trial, the data were generally in the predicted direction. It may be that we didn't study a large enough group of women to detect the difference in treatment approaches," he says. "So right now, I would not make a strong statement that there is a benefit of longer pre-quit varenicline for women, but there is enough evidence to further investigate that possibility.
"That's the likely direction we'll take next."
Hawk says there is no better approved monotherapy for smoking cessation than varenicline, which patients begin using one week before the date they are set to stop smoking.
Two preliminary clinical trials suggested that enhanced preloading beyond that one-week period could improve the drug's effectiveness. Those trials, however, used small samples and relied on self-reporting or lenient biological measures to confirm smoking abstinence.
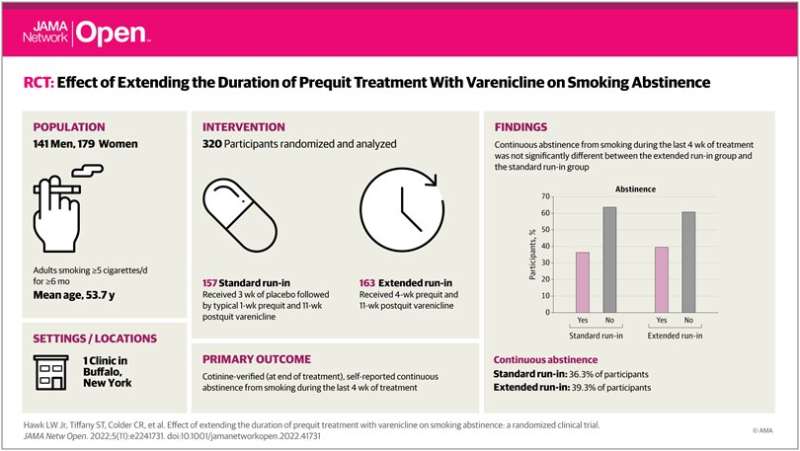
The current study more rigorously tested the hypothesis that a longer run-in (the duration the medication is taken prior to actually quitting) would improve quit rates. The researchers used a larger sample of 320 adult cigarette smokers to see if taking varenicline for more than one week prior to quitting would improve abstinence rates, measured in this case through strict bioverification.
Roughly half of the study participants received the standard one-week varenicline (with three weeks of a placebo) run-in prior to quitting; the other half received an extended four-week run-in. All the participants received brief behavioral counseling sessions focused on quit motivation, managing varenicline's side effects, smoking triggers and relapse prevention.
"When we first saw these outcomes, the team was disappointed that the study did not turn out to suggest an even more promising smoking cessation treatment," says Hawk. "But the reason for conducting research is to provide evidence. That's what sets this apart.
"We always follow the evidence."
More information: Larry W. Hawk et al, Effect of Extending the Duration of Prequit Treatment With Varenicline on Smoking Abstinence, JAMA Network Open (2022). DOI: 10.1001/jamanetworkopen.2022.41731



















