Enzyme inhibition promotes bone formation and curbs the development of bone metastases, study finds
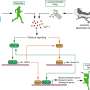
In our bones, specialized cells called osteoblasts are responsible for building up bone substance. A team of researchers led by scientists from the DKFZ-Hector Cancer Institute at the University Medical Center Mannheim and the University Medical Center Hamburg Eppendorf has now identified an enzyme that controls the activity of osteoblasts. An agent that inhibits the activity of this enzyme reduced cancer-related bone loss and the number of bone metastases in multiple myeloma and in lung and breast cancer models in mice.
Bones appear to be durable and solid. But appearances are deceptive: in fact, bone tissue is in a constant state of remodeling. Bone-degrading osteoclasts and bone-building osteoblasts ensure a fine balance in the healthy organism.
But this balance is occasionally disturbed: in osteoporosis, bone resorption takes over, so that fractures and deformities can occur. Bone metastases, which occur in the course of many cancers, are also often caused by bone resorption processes. This is also true for multiple myeloma, which originates and spreads in the bone marrow.
"Once they have penetrated the bone, many cancer cells secrete substances that suppress bone formation by osteoblasts. Patients often suffer greatly from painful bone metastases and fractures frequently occur," says Sonja Loges from the DKFZ-Hector Cancer Institute at the University Medical Center Mannheim.
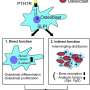
So far, drugs are available that inhibit bone resorption by osteoclasts. However, Loges and her colleague Isabel Ben Batalla believe that agents that promote bone formation by osteoblasts are also medically necessary. To identify such substances, the researchers first had to find out which signaling pathways control osteoblast activity.
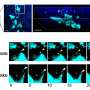
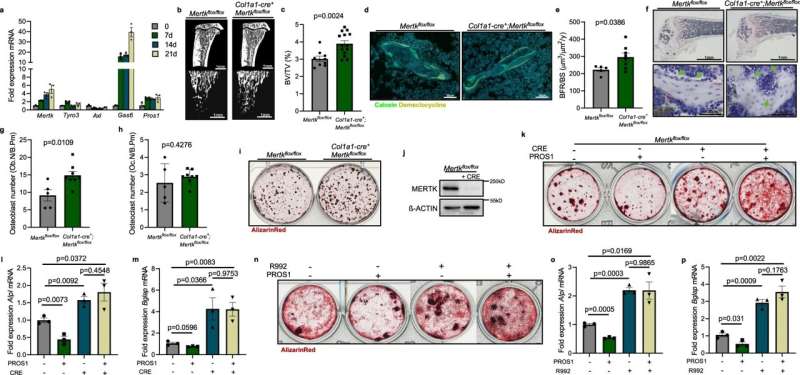
In this investigation, the team identified in mouse osteoblasts the two enzymes MERTK and Typo3, so-called receptor tyrosine kinases, which regulate bone production. The function of the two enzymes was studied in mice in whose osteoblasts either one or the other receptor tyrosine kinase was genetically switched off. The result: If MERTK was inactivated, the bone mass of the animals increased. Without Typo3, on the other hand, it decreased.
This result was an indication that the activity of MERTK in osteoblasts could also contribute to the cancer-related inhibition of bone formation.
MERTK inhibitor boosts bone formation
The small-molecule agent R992 inhibits MERTK activity. "R992 gave us a tool to test whether inhibiting MERTK could slow cancer-induced bone loss," says Janik Engelmann, first author of the study from the University Medical Center Hamburg-Eppendorf and the DKFZ-Hector Cancer Institute.
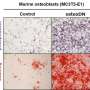
When healthy mice were treated with R992, their osteoblast numbers increased and the animals' bone mass increased. Treatment with R992 also reduced bone loss and the number of bone metastases in mouse models with myeloma, lung cancer and breast cancer cell lines.
The agent R992 is not approved as a drug. To potentially study the effects of MERTK blockade in patients, Sonja Loges' team at the German Cancer Research Center is currently developing an antibody that specifically blocks the function of MERTK. "Bone metastases affect a great many cancer patients. Osteoporosis is also a common disease. A drug that counteracts the fatal bone loss could therefore benefit a great many sufferers. We are therefore investing in further research into the role of MERTK as a therapeutic target in pathological bone loss."
The findings are published in the journal Nature Communications.
More information: Janik Engelmann et al, Regulation of bone homeostasis by MERTK and TYRO3, Nature Communications (2022). DOI: 10.1038/s41467-022-33938-x