Nanobody treatment shows promise against SARS-CoV-2 infection
A nanobody—an alternative class of antibody derived from alpaca or IIama protein fragments—developed by MassBiologics of UMass Chan Medical School, blocks SARS-CoV-2 infection in animal models and may be a new therapeutic in the growing number of biologics being explored to treat emerging COVID variants. As published in Frontiers in Immunology, a nasally delivered nanobody could be a noninvasive, low-cost and effective prophylaxis and post-exposure treatment for SARS-CoV-2 and other respiratory pathogens.
A division of UMass Chan, MassBiologics is the only nonprofit, FDA-licensed manufacturer of vaccines in the United States and is a pioneer in the development of human monoclonal antibody treatments for diseases important to public health. A 2020 study by MassBiologics was the first to show that an IgA antibody could successfully block SARS-Cov-2 infection. Nature Communications recently selected this work as one of the most impactful and innovative studies published in the journal since the COVID-19 pandemic.
"There is still a great need for therapeutics to counter emerging SARS-CoV-2 variants that cause COVID, particularly for resource-constrained populations," said principal investigator Yang Wang, MD, Ph.D., professor of medicine and deputy director of product discovery at MassBiologics.
"The challenge for antibody treatments for COVID, however, is that they have been very expensive to deliver because they have to be produced in mammalian cells and they take trained health professionals to administer intravenously. Even the mRNA vaccines require special storage and handling. Our nanobodies, in contrast, can be produced inexpensively in yeast cells and can be delivered simply in a nasal spray."
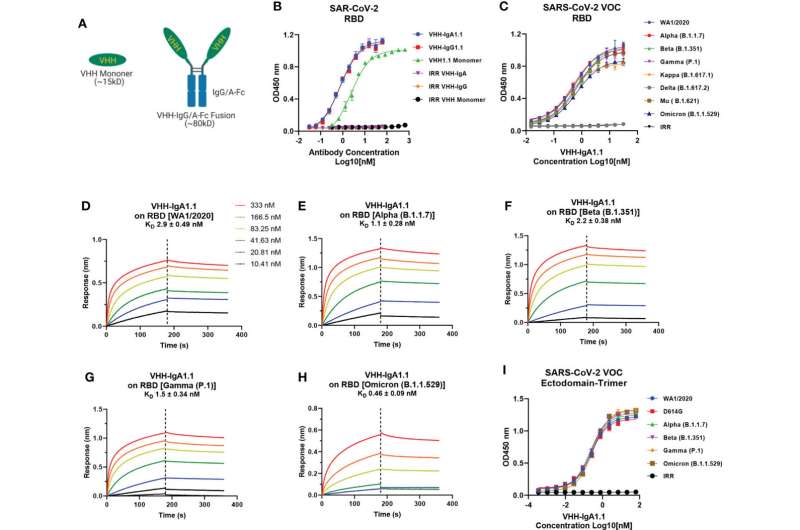
The MassBiologics nanobody, designated VHH-IgA, is a fusion of an antigen-binding fragment of the COVID spike protein and a human IgA antibody. When delivered nasally to animal models that had been previously exposed to the SARS-CoV-2 virus, the VHH-IgA effectively bonded to the SARS-CoV-2 spike protein, thereby inhibiting infection. These animals showed a marked reduction in viral loads.
Conversely, when treated prophylactically, animal models that were subsequently exposed to the SARS-CoV-2 virus showed lower levels of viral penetration, making the treatment potentially protective against COVID-19.
The VHH-IgA antibody treatment was equally effective against most variants of SARS-CoV-2, including the newer omicron variants. This suggests that a nanobody treatment may also be effective against emerging variants of concern.
"While the current mRNA vaccines and other monoclonal antibody treatments have been effective in curtailing COVID infection, there are multiple reasons why these options might not be readily available to all patients," said Dr. Wang. "It's necessary to have other options available, and our VHH-IgA molecule shows great promise as a complementary therapeutic that can neutralize existing infections and preventing new infections."
The next step for the team is to perform pre-clinical studies of the VHH-IgA biologic along with toxicology studies before embarking on clinical trials.
"Ultimately, our hope is to have a product that you can walk into and purchase, over-the-counter, from your local pharmacy," said Wang.
More information: Qi Li et al, Mucosal nanobody IgA as inhalable and affordable prophylactic and therapeutic treatment against SARS-CoV-2 and emerging variants, Frontiers in Immunology (2022). DOI: 10.3389/fimmu.2022.995412