This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Clinical trial shows significant disease-free survival for patients diagnosed with non-small cell lung cancer
New data from a Yale Cancer Center-led clinical trial shows improved rates of survival and reduced risk of recurrence in patients taking osimertinib, a targeted therapy for non-small cell lung cancer (NSCLC). NSCLC is the most common type of lung cancer, and for patients diagnosed with more advanced disease stages, cases tend to recur.
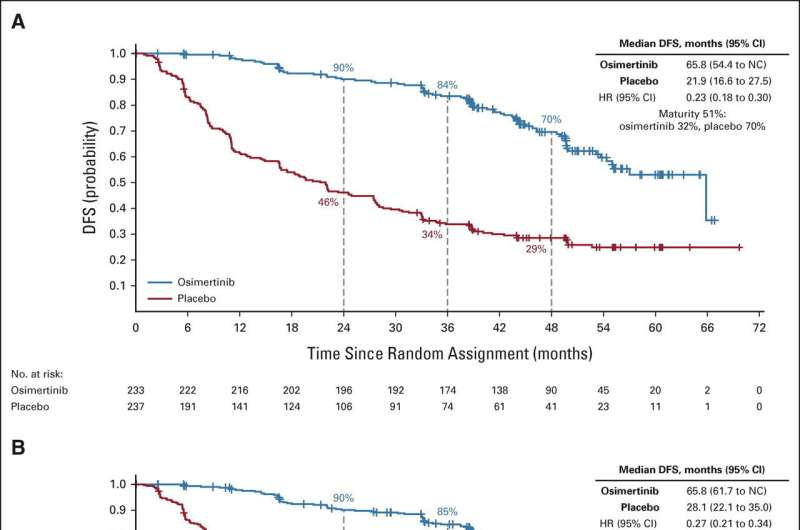
The phase III ADAURA clinical trial assessed the safety and efficacy of osimertinib in patients with surgically removed (completely resected) stage IB-IIA NSCLC, who were previously treated with or without adjuvant chemotherapy. The updated trial results were published in the Journal of Clinical Oncology on January 31.
The new data show there are ongoing benefits for patients with NSCLC taking osimertinib, including prolonged disease-free survival (DFS) over placebo, reduced risk of local and distant metastases (spread of tumor), and improved central nervous system DFS. These findings support the efficacy of adjuvant osimertinib in the patient population studied during the trial, researchers say.
"These data demonstrate a new paradigm demonstrating the importance of using targeted therapy against epidermal growth-factor-driven tumors as early as possible in the course of a patient's disease," said Dr. Roy S. Herbst, lead author and principal investigator of the trial, ensign professor of medicine (medical oncology) and professor of pharmacology and deputy director at Yale Cancer Center. "The results have led to a new standard of care in this disease setting."
Herbst is also chief of medical oncology and director of the Center for Thoracic Cancers at Smilow Cancer Hospital and the Yale Cancer Center, and assistant dean for translational research at Yale School of Medicine.
The trial enrolled 682 patients with stage IB-IIA NSCLC, who were randomly assigned to receive 80 milligrams of osimertinib once daily or a placebo. Herbst and his research team found that fewer patients treated with osimertinib experience recurrence. Data also showed that fewer patients had distant metastases when compared to the placebo group.
The found that disease-free survival (DFS) over four years was 73% for the osimertinib group and 38% for the placebo group. And fewer patients who were randomly assigned osimertinib had disease recurrence (27%) than those in the placebo group (60%). The data from this study demonstrate prolonged DFS and reduced local- and distant-recurrence of symptoms, supporting osimertinib as a highly effective treatment in patients with resected EGFR-mutated stage IB-IIA non-small cell lung cancer.
"This therapy was well tolerated and prevented patients from developing metastasis to distant sites such as the brain, bone, and other areas of the lungs," Herbst said. "This has truly impacted the lives of our patients."
The study was supported by AstraZeneca, the manufacturer of osimertinib.
More information: Roy S. Herbst et al, Adjuvant Osimertinib for Resected EGFR-Mutated Stage IB-IIIA Non–Small-Cell Lung Cancer: Updated Results From the Phase III Randomized ADAURA Trial, Journal of Clinical Oncology (2023). DOI: 10.1200/JCO.22.02186