This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Researchers make gains in race to protect against RSV
As a surge in cases of respiratory syncytial virus, or RSV, continues pushing hospitals to capacity across the U.S., researchers at Georgia State's Institute for Biomedical Sciences are working to develop vaccines and other forms of treatment to fight this and other serious respiratory viruses.
RSV is a common virus that usually causes mild, cold-like symptoms in adults, but the past two years have seen a dramatic rise in unseasonably early cases that can be life-threatening to infants and older adults. It's also a leading cause of bronchiolitis and pneumonia in children less than 1 year old, according to the National Institutes of Health (NIH).
According to the Atlanta-based Centers for Disease Control and Prevention (CDC), RSV cases recently hit a two-year high in the U.S. Leaders in Orange County, Calif., declared a public health emergency over the surge in cases. In Detroit, Mich., and elsewhere, hospitals are dealing with a shortage of amoxicillin because of soaring case numbers.
"As with coronavirus, we are seeing other dangerous viruses like RSV change and spread in unexpected ways," said Tim Denning, vice president for research and economic development at Georgia State. "That's why it's so crucial to understand how these viruses evolve and how to best protect all groups, including those who are most vulnerable."
Distinguished University Professor and Director of the Center for Translational Antiviral Research Richard Plemper has developed an oral antiviral drug that targets a key part of the RSV polymerase and inhibits the synthesis of viral genetic material, a finding that could provide an effective treatment against RSV disease.
"In 2022 some very promising new drug candidates for the treatment of RSV disease have been released," said Plemper, "and we are excited to contribute with Georgia State's clinical candidate to this important development."
Plemper is co-founder of the newly established Antiviral Countermeasures Development Center (AC/DC). It's one of nine Antiviral Drug Discovery (AViDD) Centers around the country funded by the National Institute of Allergy and Infectious Diseases (NIAID).
This new collaborative center at Emory University and Georgia State is being led by Plemper and co-founder George Painter of Emory. The researchers are world-renowned for their work developing successful antiviral drugs, including molnupiravir, one of the world's first antiviral pills approved for use against SARS-CoV-2. Their previous collaborations have involved drugs to treat RSV as well as other paramyxoviruses that cause mumps and measles.
Sang-Moo Kang's research is based around the design and development of effective vaccines against viral diseases such as influenza virus and RSV, better understanding vaccine-induced protective immune mechanisms, studying pathogen-induced inflammation and developing anti-inflammatory therapeutics.
"We are focusing on a new nanoparticulate vaccine retaining native-like RSV fusion protein to effectively induce neutralizing antibodies blocking virus entry into the host cells, and possible dual vaccines against flu and RSV in future projects by utilizing mRNA and nanoparticle platforms," Kang said.
The research is significant for the development of effective and universal flu vaccines, as well as development of a vaccine against RSV, which does not currently exist. Kang's work also involves understanding neonatal immunology, vaccine-induced immunity and protective immune mechanisms and serious inflammatory pathogens, among other applications.
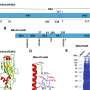
A new U.S. patent is being issued based on one of Kang's discoveries. It focuses on RSV vaccine constructs molecularly designed and genetically engineered to comprise RSV pre-fusion proteins displayed on the surface of a particle. The research was conducted with the support of the NIH.
Kang is also focused on the creation of a unique adjuvant, a chemical added to a vaccine to prime the immune system, that prevents lung inflammation.
Cliff Michaels, assistant vice president and director of Georgia State's Office of Technology Transfer & Commercialization said these are the types of advances that can have real-world impact. "The pandemic has clearly reminded us that respiratory viral infections are to be taken seriously and our continued research and collaboration with academic and industry partners is critical to bringing these discoveries to the public," Michaels said.