This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Study: Senescence-associated exosomes transfer fibrosis to neighboring cells
A new research paper, titled "Senescence-associated exosomes transfer miRNA-induced fibrosis to neighboring cells," was published in Aging.
Radiation-induced fibrosis is a common side effect of radiotherapy, which is the most common treatment for cancer. However, radiation also causes p53-mediated cell cycle arrest, prolonged expression of p21, and the development of senescence in normal cells that reside in irradiated tissues. Bone marrow-derived mesenchymal stem cells (MSCs) accumulate in primary tumor sites because of their natural tropism for inflammatory and fibrotic tissues.
MSCs are extremely sensitive to low doses of ionizing radiation and acquire senescence as a result of bystander radiation effects. Senescent cells remain metabolically active but develop a potent senescence-associated secretory phenotype (SASP) that correlates to hyperactive secretion of cytokines, pro-fibrotic growth factors, and exosomes (EXOs).
Integrative pathway analysis has highlighted that radiation-induced senescence significantly enriched cell-cycle, extracellular matrix, transforming growth factor-β (TGF-β) signaling, and vesicle-mediated transport genes in MSCs. EXOs are cell-secreted nanovesicles (a subclass of small extracellular vesicles) that contain biomaterials—proteins, RNAs, microRNAs (miRNAs)—that are critical in cell-cell communication. miRNA content analysis of secreted EXOs further revealed that radiation-induced senescence uniquely altered miRNA profiles.
"In fact, several of the standout miRNAs directly targeted TGF-β or downstream genes," write the researchers.
In this new study, researchers Amy H. Lee, Deepraj Ghosh, Ivy L. Koh, and Michelle R. Dawson from Brown University further treated normal MSCs with senescence-associated EXOs (SA-EXOs) to examine bystander effects of radiation-induced senescence.
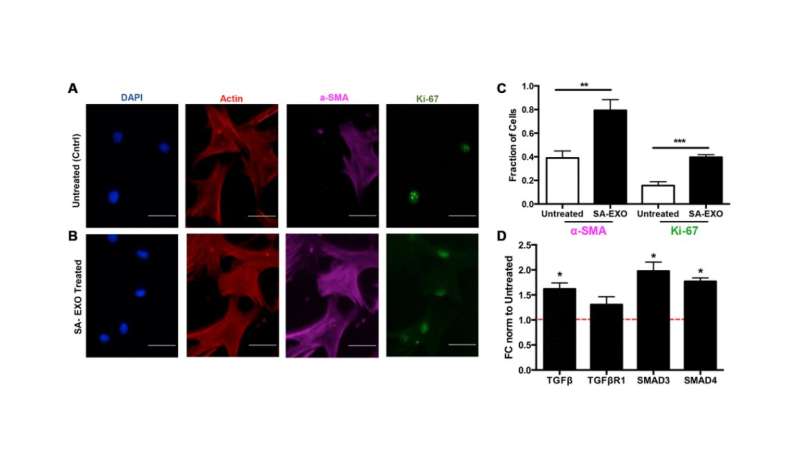
The researchers found that these modulated genes were related to TGF-β pathway and elevated both alpha smooth muscle actin (protein increased in senescent, activated cells) and Ki-67 (proliferative marker) expression in SA-EXO treated MSCs compared to untreated MSCs. They revealed that SA-EXOs possess unique miRNA content that influence myofibroblast phenotypes via TGF-β pathway activation. This highlights that SA-EXOs are potent SASP factors that play a large role in cancer-related fibrosis.
"Our integrated omics and EXO microarray analyses show that senescent MSCs possess differential transcriptional genes and secrete vesicles that contain unique post-transcriptional cargo. We subsequently demonstrated that these EXO miRNAs can play important roles in cell-cell communication during disease progression," conclude the authors.
More information: Amy H. Lee et al, Senescence-associated exosomes transfer miRNA-induced fibrosis to neighboring cells, Aging (2023). DOI: 10.18632/aging.204539