This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New approach targets norovirus, world's leading cause of foodborne infection
Every year, norovirus causes hundreds of millions of cases of food poisoning—and the deaths of at least 50,000 children—yet there exists no real way to control it. The virus has proven exceptionally difficult to study in the lab, and scientists have struggled to develop effective vaccines and drugs.
A new study at Washington University School of Medicine in St. Louis describes a creative way to make a vaccine against norovirus by piggybacking on the highly effective vaccines for rotavirus, an unrelated virus that also causes diarrhea.
The researchers created an experimental rotavirus-norovirus combo vaccine by adding a key protein from norovirus to a harmless strain of rotavirus. Mice that received the experimental vaccine produced neutralizing antibodies against both rotavirus and norovirus. The study, available online in Proceedings of the National Academy of Sciences, outlines an innovative approach to preventing one of the most common and intractable viral infections.
"Pretty much everyone has had norovirus at some point," said senior author Siyuan Ding, Ph.D., an assistant professor of molecular microbiology. "You go out to eat, and the next thing you know you're vomiting and having diarrhea. You will recover, but it's going to be a rough three days or so. For kids in the developing world who don't have access to clean water, though, it can be deadly. The rotavirus vaccines work really well, and there are already global distribution systems set up for them, so based on that, we saw an opportunity to finally make some headway against norovirus."
Before the first rotavirus vaccines were rolled out in 2006, half a million children around the world died every year of diarrhea caused by rotavirus infection. Now, the number is estimated to be about 200,000—still high but a huge improvement. Four rotavirus vaccines are in use around the world. All are live-virus vaccines, meaning they are based on weakened forms of rotavirus capable of triggering an immune response but not of making people sick.
Human norovirus, on the other hand, has stymied scientific investigation for decades. It doesn't infect mice or rats or any other ordinary lab animals, so the kinds of experiments that led to the development of rotavirus vaccines have been impossible to replicate with norovirus.
Ding and colleagues—including first author Takahiro Kawagishi, Ph.D., a staff scientist in Ding's lab, and co-corresponding author Harry B. Greenberg, MD, a professor emeritus of medicine at Stanford University—came up with the idea of using rotavirus to bypass the technical difficulties of working with norovirus. They worked with a laboratory strain of rotavirus as a stand-in for one of the approved rotavirus vaccines, which are proprietary.
The researchers inserted the gene for the protein that forms the outer surface of human norovirus into the genome of the rotavirus lab strain. Then, they administered the modified rotavirus to immunocompromised infant mice by mouth, the same way rotavirus vaccines are given to children. They took blood and fecal samples four, six and eight weeks later. Nine weeks after the initial immunization, the researchers gave the mice a booster by injection and took samples again a week later.
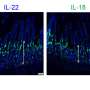
A strong antibody response was evident in the blood of nine of 11 mice tested, and in the intestines of all 11 mice. Even better, some of the antibodies from the blood and the intestines were able to neutralize both viruses in human "mini-gut" cultures in a dish. Such cultures, also known as organoids, are grown from human stem cells and replicate the surface of the human gut.
"Traditionally, vaccine studies have focused on the antibody response in the blood, because we understand that part of the immune response the best," Ding said. "But norovirus and rotavirus are gut viruses, so antibodies in the blood are less important than the ones in the intestines in terms of fighting off these viruses. The fact that we saw a strong antibody response in the intestines is a good sign."
The next step is to show that animals immunized with the experimental vaccine are less likely to get sick or die from norovirus. Ding has such experiments underway.
The power of this study is that it outlines a novel approach that could accelerate vaccine development for a variety of troublesome organisms that cause diarrhea, especially in resource-limited countries where many of these infections occur.
"There are a lot of intestinal pathogens out there for which we don't have good treatments or vaccines," Ding said. "In principle, we could put a gene from any organism that infects the intestinal tract into the rotavirus vaccine to create a bivalent vaccine. We'd have to find the right targets to produce a good immune response, of course, but the principle is simple.
"As basic scientists, we rarely get the chance to actually move something forward into the clinic," Ding continued. "We study what the virus does and how the host responds at a basic level. This is a rare opportunity for our work to affect human health directly and make people's lives better."
More information: Takahiro Kawagishi et al, Mucosal and systemic neutralizing antibodies to norovirus induced in infant mice orally inoculated with recombinant rotaviruses, Proceedings of the National Academy of Sciences (2023). DOI: 10.1073/pnas.2214421120