This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Protein Beclin-1 shown to be a major player in uterine remodeling and the establishment of pregnancy
Throughout a woman's reproductive life, the endometrium, the mucous membrane lining the uterus, goes through cyclical remodeling. It thickens during the menstrual cycle in preparation for embryo implantation, and it is shed during menstruation when there is no fertilization.
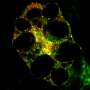
Researchers at Baylor College of Medicine and collaborating institutions are investigating little-known factors directing uterine remodeling to advance the understanding of this process and provide new insights into fertility-associated gynecological conditions. They report today (April 10) in the journal Developmental Cell that autophagy protein Beclin-1 governs endometrial remodeling and the establishment of pregnancy in animal models by regulating autophagy, a natural process that digests and recycles cellular components.
"One of our main goals is to understand biological processes that might be associated with pregnancy loss," said corresponding author Dr. Rama Kommagani, associate professor in the Departments of Pathology and Immunology and Molecular Virology and Microbiology at Baylor. "In this study, we looked to understand the role Beclin-1 plays in endometrial reprogramming for the successful establishment of pregnancy."
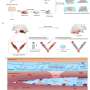
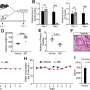
In the first part of this study, Kommagani and his colleagues found that Beclin-1 is essential for the establishment of pregnancy in mouse models. When they removed Beclin-1 specifically from uterine cells, the uteri did not develop properly, and this led to infertility caused by reduced uterine receptivity and failed embryo implantation.
In addition, molecular analysis of uteri revealed that Beclin-1 is necessary for endometrial programming of stem cells that leads to the development of uterine glands and other structures that are essential for uterine maturation.
"We also studied the mechanism by which Beclin-1 regulates uterine development," Kommagani said. "Beclin-1 is known to regulate two distinct natural processes called autophagy and apoptosis that help maintain many normal functions in organisms. We investigated whether one of these two processes or both were involved."
The team found that disabling apoptosis in the presence of Beclin-1did not alter the normal remodeling of the uterus and gland development. Importantly, restoring only Beclin-1-driven autophagy, but not apoptosis, promoted normal uterine remodeling.
"After birth, the uterus goes through a multistep process of development that is required for successful pregnancies in the future. Any deficiencies along this process can have a ripple effect on the ability to conceive and for the pregnancy to succeed," Kommagani said. "We discovered that one of the steps involves Beclin-1-mediated autophagy, which acts as a molecular switch that governs uterine reprograming by maintaining the endometrial progenitor stem cells that support uterine remodeling."
The findings have encouraged the researchers to explore the possibility of developing practical applications both in the field of contraceptives and infertility.
More information: Vojo Kommagani, Beclin-1-Dependent Autophagy but Not Apoptosis Is Critical for Stem Cell Mediated-Endometrial Programming and the Establishment of Pregnancy, Developmental Cell (2023). DOI: 10.1016/j.devcel.2023.03.013. www.cell.com/developmental-cel … 1534-5807(23)00109-0