This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Gut microbiome and its products promote endometriosis in animal model
About 196 million women worldwide suffer from endometriosis, a condition that typically causes pelvic pain and infertility. Endometriosis develops when lining inside the womb grows attached to surrounding tissues, such as the intestine or the membrane lining the abdominal cavity, causing bleeding, pain and other symptoms. Despite decades of research, little is known about the factors that contribute to the development of endometriosis.
Evidence suggests that the microbiome, a community of microorganisms living inside the body, is altered in women with endometriosis. In this study published in the journal Cell Death & Discovery, researchers at Baylor College of Medicine discovered that an altered gut microbiome plays a pivotal role in endometriosis disease progression in an animal model.
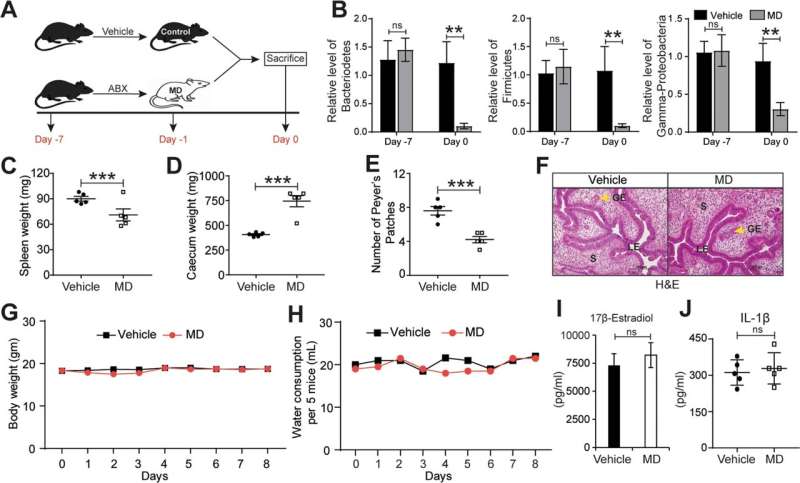
"To investigate the role of the microbiome in endometriosis we first implemented a novel mouse model of the condition in which we eliminated the microbiome using antibiotics," said lead author Dr. Rama Kommagani, associate professor in the Departments of Pathology and Immunology and of Molecular Virology and Microbiology at Baylor.
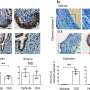
The researchers found that mice lacking gut microbiome had smaller endometriotic lesions than mice with a microbiome. Furthermore, when gut microbiome-free mice received gut microbiota from mice with endometriosis, the lesions grew as large as those in mice retaining their microbiome. These findings suggest that altered gut bacteria drive disease progression. On the other hand, the uterine microbiome did not seem to affect disease progression.
The team also discovered a novel signature of microbiome-derived metabolites, products produced by the microbes, that were significantly altered in feces of mice with endometriosis. Supporting the role of microbiome metabolites in disease progression, Kommagani and his colleagues found that treatment of endometriotic cells and mice with the metabolite called quinic acid significantly enhanced the cellular proliferation and endometriotic lesion growth, respectively.
The findings suggest that certain microbiome communities and/or their metabolites can contribute to endometriosis progression and that modifying the composition of these communities could help control the condition in human patients. "We are currently investigating this possibility," Kommagani said.
The findings also suggested that studying microbiome metabolites in human stool samples could be used as a diagnostic tool. "Endometriosis is typically diagnosed with ultrasound, and an invasive procedure is necessary to characterize the lesion well," Kommagani said. "We are investigating whether microbiome metabolites in human stool samples could be a useful diagnostic tool and also whether some of these metabolites could be used as a treatment strategy."
Women with endometriosis also tend to have bowel issues, such as colitis or inflammatory bowel syndrome. "We are interested in determining whether changes in the gut microbiome could affect bowel conditions and the possibility of controlling them by modifying the microbiome or with their metabolites," Kommagani said.
More information: Sangappa B. Chadchan et al, Gut microbiota and microbiota-derived metabolites promotes endometriosis, Cell Death Discovery (2023). DOI: 10.1038/s41420-023-01309-0