This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Using AI to predict an important measure of heart performance
Coronary heart disease is the leading cause of adult death worldwide. The coronary angiography procedure provides the clinical standard diagnostic assessment for nearly all related clinical decision-making, from medications to coronary bypass surgery. In many cases, quantifying left ventricular ejection fraction (LVEF) at the time of coronary angiography is critical to optimize clinical decision-making and treatment decisions, particularly when angiography is performed for potentially life-threatening acute coronary syndromes (ACS).
Since the left ventricle is the heart's pumping center, measuring the ejection fraction in the chamber provides critical information about the percentage of blood leaving the heart each time it contracts. Presently, measuring LVEF during angiography requires an additional invasive procedure called left ventriculography—where a catheter is inserted into the left ventricle and contrast dye is injected—which carries additional risks and increases the contrast exposure.
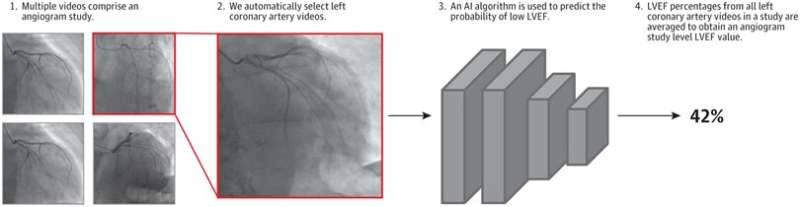
In a study publishing in JAMA Cardiology, senior author and UCSF cardiologist Geoff Tison, MD, MPH, and first author Robert Avram, MD, of the Montreal Heart Institute, set out to determine whether deep neural networks (DNNs), a category of AI algorithm, could be used to predict cardiac pump (contractile) function from standard angiogram videos. They developed and tested a DNN called CathEF, to estimate LVEF from coronary angiograms of the left side of the heart.
"CathEF offers a novel approach that leverages data that is routinely collected during every angiogram to provide information that is not currently available to clinicians during angiography, effectively expanding the utility of medical data with AI and provides real-time LVEF information that informs clinical decision-making," said Tison, UCSF associate professor of Medicine and Cardiology.
The researchers performed a cross-sectional study of 4,042 adult angiograms matched with corresponding transthoracic echocardiograms (TTEs) from 3,679 UCSF patients and trained a video-based neural network to estimate reduced LVEF (less than or equal to 40%) and to predict (continuous) LVEF percentage from standard angiogram videos of the left coronary artery.
The results showed that CathEF accurately predicted LVEF, with strong correlations to echocardiographic LVEF measurements, the standard noninvasive clinical approach. The model was also externally validated in real-world angiograms from the Ottawa Heart Institute. The algorithm performed well across different patient demographics and clinical conditions, including acute coronary syndromes and varying levels of renal function—patient populations that may be less well suited to receive the standard left ventriculogram procedure.
"This study presents a novel method for assessing LVEF, an important measure of heart function, during any routine coronary angiography without requiring additional procedures or increasing cost," said Avram, an interventional cardiologist and former UCSF research fellow. "LVEF is essential for making decisions during the procedure and for managing patient care."
Although the algorithm was trained on a large dataset of angiograms from UCSF and then separately validated in a dataset from the Ottawa Heart Institute, the investigators are undertaking further research to test this algorithm at the point-of-care and determine its impact on the clinical workflow in patients suffering heart attacks. To this end, a multi-center prospective validation study in patients with ACS is underway to compare the performance of CathEF and the left ventriculogram with TTEs performed within 7 days of ACS.
"This work demonstrates that AI technology has the potential to reduce the need for invasive testing and improve the diagnostic capabilities of cardiologists, ultimately improving patient outcomes and quality of life," said Tison.
More information: Automated Assessment of Cardiac Systolic Function From Coronary Angiograms With Video-Based Artificial Intelligence Algorithms, JAMA Cardiology (2023). DOI: 10.1001/jamacardio.2023.0968