This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Study: Blood vessel damage could be an Alzheimer's driver
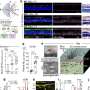
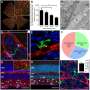
![Loss of retinal endothelial claudin-5 in MCI and AD patients in relation to retinal vascular amyloidosis, CAA, and cognitive deficit. A, Representative images of immunofluorescent staining for claudin-5 (red), collagen IV (ColIV, green), and DAPI (blue) on post mortem cross-sections of retina from CN (n = 21 [control]) patients as well as from patients with MCI (n = 10) and those with AD (n = 21) with different degrees of CAA severity scores. All scale bars = 20 µm. B,C, Quantitative analysis of retinal vascular claudin-5 IR separately in capillaries and LBVs from all experimental groups stratified by (B) CAA severity scores and by (C) diagnostic groups (n = 53 in total). D, Average of retinal vascular claudin-5 IR in capillaries and LBVs stratified by sex in the same cohort (n = 53 total). E, Pearson's coefficient (r) correlation between CAA severity scores and claudin-5 in retinal capillaries (red) and LBVs (gray) (n = 35 total). F, Heatmaps illustrating Pearson's correlations between retinal claudin-5 in capillaries and LBVs versus brain pathology and cognitive decline, including Aβ plaques (Aβ-P), NFTs, NTs, ATR, MMSE scores, CDR scores, CAA severity scores, Braak stages, and A (amyloid) B (Braak) C (Consortium to Establish a Registry for Alzheimer's Disease) average scores in AD (n = 18), MCI (n = 10), and CN (n = 9) human donors (n = 37 total). Pseudo-color red and numbers demonstrate the strength of (r) correlation power; statistical significance is demonstrated as follows: n.s., not significant, *P < 0.05, **P < 0.01, ****P < 0.0001. G,H, Pearson's coefficient (r) correlation between (G) retinal Aβ40 versus claudin-5 in capillaries and (H) retinal arteriolar Aβ40 versus claudin-5 in LBVs. Data from individual donors (circles) as well as group means ± standard error of the mean are shown. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, by one-way analysis of variance with Tukey's post hoc multiple comparison test. Two-group statistical analysis was performed using an unpaired two-tailed Student t test, and results are shown in parentheses. Percentage decreases are shown in red. Aβ, amyloid beta; AD, Alzheimer's disease; ATR, atrophy; B, Black; C, Caucasian (White); CAA, cerebral amyloid angiopathy; CDR, Clinical Dementia Rating; CN, cognitively normal; F, female; H, Hispanic; IR, immunoreactivity; LBVs, large blood vessels; MCI, mild cognitive impairment; MMSE, Mini-Mental State Examination; NFTs, neurofibrillary tangles; NTs, neutrophil threads; yrs, years old. Credit: Alzheimer's & Dementia (2023). DOI: 10.1002/alz.13086 Study: Blood vessel damage could be an Alzheimer's driver](https://scx1.b-cdn.net/csz/news/800a/2023/study-blood-vessel-dam.jpg)
Blood vessel abnormalities in the eye are a major factor in the progression of Alzheimer's disease, according to research from Cedars-Sinai investigators published in the journal Alzheimer's & Dementia. These changes correspond to changes in the brain, offering a new possibility for early diagnosis.
"This study provides a new understanding of the vascular changes associated with Alzheimer's disease, especially in the retina, the layer of nerve tissue at the back of the eye," said Maya Koronyo-Hamaoui, Ph.D., professor of Neurology, Neurosurgery and Biomedical Sciences at Cedars-Sinai and senior author of the study. "It also points to the damage Alzheimer's disease causes to the blood vessels in the retina, enabling a new, noninvasive pathway to early diagnosis and monitoring of disease progression."
Investigators compared blood vessels in retinas collected from 24 human donors with Alzheimer's disease, 10 donors with mild cognitive impairment and 27 with normal cognition.
In patients with Alzheimer's disease and mild cognitive impairment, they found one of the earliest signs of Alzheimer's disease to date: disruption of the blood-retinal barrier, where tightly joined cells prevent harmful substances from entering the retinal tissue.
"In patients with Alzheimer's disease, we found that there was a deficit of as much as 70% in that barrier, meaning harmful substances can pass through and enter the retinal tissue," said Haoshen Shi, MD, Ph.D., project scientist and first author of the study. "We see that this occurs very early on, in patients with only mild functional impairment."
Damage to the blood-retinal barrier was strongly associated with a condition called cerebral amyloid angiopathy (CAA)—the accumulation of amyloid proteins in small blood vessels—and other vascular disease in the brain.
"Currently, the only way to detect CAA in patients is in post-mortem brain tissue samples," said Koronyo-Hamaoui. "With additional research and the development of advanced retinal imaging, vascular and blood-retinal barrier damage could give us the first opportunity to detect CAA in living patients."
The study also found that deposits of a protein called amyloid beta 40 accumulated in the retinal arteries of Alzheimer's disease patients, making the arteries stiff, disrupting blood flow and preventing the arteries from clearing harmful substances from the retina. Further studies are necessary to determine whether the deposits accumulate because of blood vessel damage, or actually cause the damage, Koronyo-Hamaoui said.
"Retinal and brain tissues are rich in blood vessels, and high blood supply is fundamental for their function," Koronyo-Hamaoui said. "Restriction of blood supply, which may occur due to the damage we show happening here, means that these cells do not get the oxygen and the nutrients that they need."
Advanced retinal imaging, which would look at the blood vessels and protein accumulation noninvasively in living patients, is in development but is not yet approved by the Food and Drug Administration, Koronyo-Hamaoui said.
"As an anatomical extension of the brain, the retina has been extensively examined as a window to central nervous system disorders," said Keith L. Black, MD, chair of the Department of Neurosurgery and the Ruth and Lawrence Harvey Chair in Neuroscience at Cedars-Sinai. "This work adds to recent progress in advanced retinal imaging and identification of other retinal biomarkers to advance the science of Alzheimer's disease early detection."
Meanwhile, Koronyo-Hamaoui advises people to do what they can to keep their circulatory system, including the blood vessels in the retina and the brain, healthy to help prevent CAA and dementia.
"Controlling hypertension, eating a healthy diet low in sugar, reducing alcohol consumption and avoiding smoking helps prevent chronic inflammation and damage to blood vessels," Koronyo-Hamaoui said. "Our study shows that blood vessel damage is a major element in the progression of Alzheimer's disease."
More information: Haoshen Shi et al, Retinal arterial Aβ 40 deposition is linked with tight junction loss and cerebral amyloid angiopathy in MCI and AD patients, Alzheimer's & Dementia (2023). DOI: 10.1002/alz.13086