This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Researchers report on intrathoracic synovial sarcoma with BRAF V600E mutation
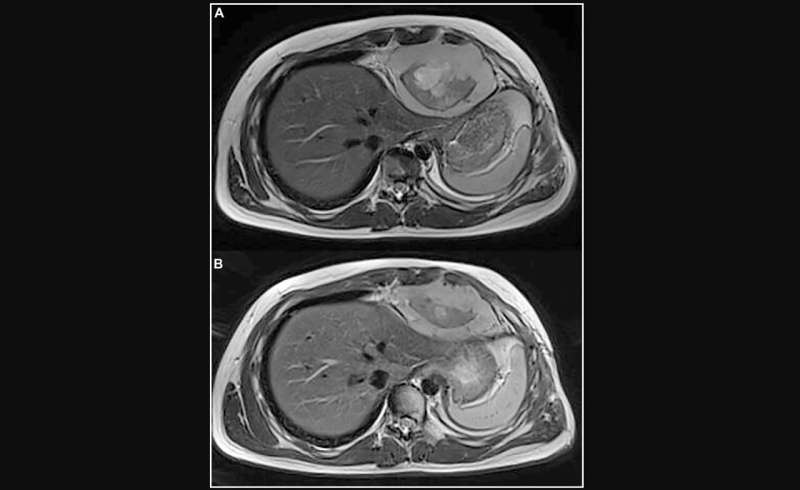
A new case report titled "Intrathoracic synovial sarcoma with BRAF V600E mutation" has been published in Oncotarget.
Synovial sarcoma (SS) is a highly malignant mesenchymal tumor that occurs mainly in adolescents and young adults. The treatment of SS is multimodal, involving surgery, radiotherapy and chemotherapy. The overall prognosis is generally quite satisfactory in children and adolescents with localized SS at diagnosis. However, the outcome remains poor for patients who relapse, with a reported 5-year post-relapse survival of around 30%.
In this new paper, researchers Ida Russo, Sabina Barresi, Pier Luigi Di Paolo, Valentina Di Ruscio, Giada Del Baldo, Annalisa Serra, Silvia Vallese, Evelina Miele, Angela Mastronuzzi, Rita Alaggio, Andrea Ferrari, and Giuseppe Maria Milano from Italy's Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) report the case of a 15-year-old boy with intrathoracic synovial sarcoma who relapsed after standard chemotherapy, surgery and radiotherapy. The molecular analysis of the tumor identified a BRAF V600E mutation at time of progression of relapsed disease under third-line systemic treatment.
This mutation is commonly seen in melanomas and papillary thyroid cancers, but less prevalent (typically less than 5%) across a variety of other cancer types. The patient underwent selective BRAF inhibitor vemurafenib treatment achieving partial response (PR) with a progression-free survival (PFS) ratio of 1.6 months and an overall survival of 19 months, alive in continuous PR. This case highlights the role of routine next-generation sequencing (NGS) used to drive treatment choice and to investigate SS tumors for BRAF mutations.
"Our data highlight the importance of implementing molecular tests in SS patients to evaluate BRAF mutational actual incidence in these neoplasms," the researchers conclude.
More information: Ida Russo et al, Intrathoracic synovial sarcoma with BRAF V600E mutation, Oncotarget (2023). DOI: 10.18632/oncotarget.28475


















