This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New rapid test uses bacteriophages to identify pathogens that cause urinary tract infections
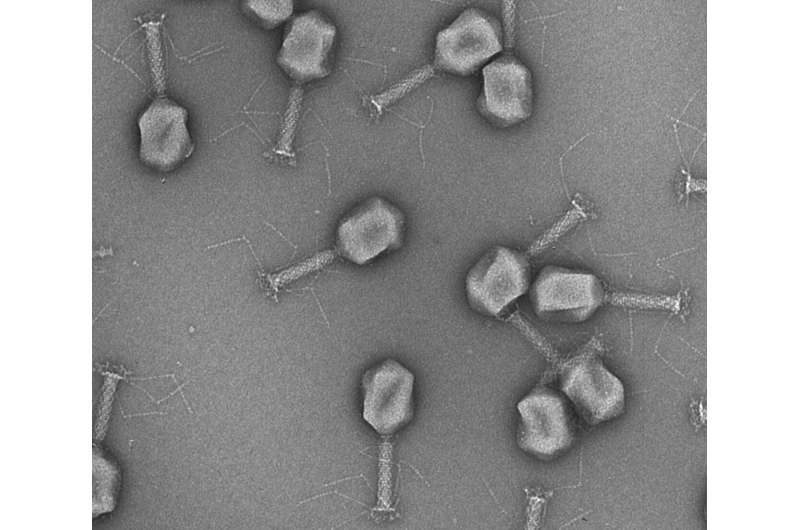
About one in two women are affected by cystitis during their lifetime, and many suffer from recurrent urinary tract infections. Bladder infections are not only painful and potentially dangerous, but they also pose a significant dilemma for physicians.
With antibiotic resistance becoming widespread in urinary tract infections and continually increasing, physicians are often forced to blindly prescribe antibiotics without knowing their effectiveness against the pathogen causing the infection. This is because it takes several days to identify a specific pathogen using conventional diagnostics.
Researchers at ETH Zurich, in collaboration with Balgrist University Hospital, have now developed a rapid test that employs the natural viral predators of bacteria, bacteriophages. The researchers also genetically modified the phages to make them more efficient at destroying the pathogenic bacteria. The research has been published in Nature Communications.
Fast and reliable diagnosis
Phages are highly specialized viruses. Each species of phage infects only one particular type or strain of bacteria. ETH Zurich scientists from the Food Microbiology research group led by Professor Martin Loessner are now taking advantage of this unique characteristic.
The first step was to identify the phages that are effective against the three main types of bacteria implicated in urinary tract infections, namely Escherichia coli, Klebsiella and Enterococci. These natural phages were then modified in such a way that any bacteria they recognize and infect are propelled to produce an easy-to-measure light signal.
Using this method, the researchers were able to reliably detect the pathogenic bacteria directly from a urine sample in less than four hours. In the future, the method could make it possible to prescribe a suitable antibiotic immediately after diagnosis and thus minimize resistance development and improve antibiotic stewardship.
The method also has another advantage: it allows physicians to predict which patients are likely to respond particularly well to a tailored phage therapy, as the strength of the light signal produced in the assay already indicates how efficient the phages are in attacking the bacterium—the more the sample glows, the better the bacterium will respond to the therapy.
Double-action sniper
Phage therapies have been used for over 100 years but fell into oblivion in Western industrialized countries with the discovery of penicillin. In view of increasing antibiotic resistance, they are currently seeing a renaissance. They also have the decisive advantage of attacking only a single target bacterium, much like a sniper.
However, previous therapeutic approaches have one problem, "Phages aren't interested in completely killing their host, the pathogenic bacterium," explains ETH researcher Samuel Kilcher, one of the study's two final authors.
To enhance the phages' effectiveness, the researchers genetically modified them. The modified phages produce not only new phages inside the infected host bacterium, but also bacteriocins. Once they are released, these bacteria-killing proteins are particularly effective against bacterial strains that have altered parts of their surface in such a way that the phages no longer recognize them. This double-barrelled attack makes the treatment more effective.
From the laboratory to the clinic
In individual cases, such as the recent rescue of a lung patient at the University Hospital of Geneva, phage therapies have been successfully used experimentally.
"There are also many academic and commercial clinical trials underway worldwide that are systematically investigating the potential of natural and genetically optimized phages," says Matthew Dunne, one of the study's final authors. However, there is a long way to go before such therapies can be applied more widely in Western countries. In addition to extensive clinical studies, regulatory adjustments would also be useful, taking into account the fact that phages are biological entities that co-evolve with their bacterial hosts, i.e., they are constantly evolving.
The present study is a proof of concept. Next, the ETH Zurich researchers, together with their partners from Balgrist University Hospital, will test the efficacy of the new phage therapy in a clinical trial with selected patients.
More information: Jiemin Du et al, Enhancing bacteriophage therapeutics through in situ production and release of heterologous antimicrobial effectors, Nature Communications (2023). DOI: 10.1038/s41467-023-39612-0
Susanne Meile et al, Engineered reporter phages for detection of Escherichia coli, Enterococcus, and Klebsiella in urine, Nature Communications (2023). DOI: 10.1038/s41467-023-39863-x