This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Seroprevalence of human enterovirus A71 in Guangzhou, China, 2019–2021
Enterovirus A71 (EV-A71) is a significant hand-foot-mouth disease (HFMD) etiology. The inactivated EV-A71 vaccines were approved in China in 2016. However, the seroprevalence of EV-A71 after the vaccine application and its potential association with the EV-A71 epidemic in the population have been rarely studied.
The authors of an article published in Biosafety and Health analyzed the incidence of EV-A71 infection and seroepidemiology in Guangzhou City, China. From 2019 to 2021, 167,920 clinically confirmed HFMD cases were reported in Guangzhou. In 6,868 enterovirus-positive samples, Coxsackievirus A6 and Coxsackievirus A16 were dominant genotypes, and only three EV-A71-positive samples were detected, highlighting the deficient epidemic activity of EV-A71.
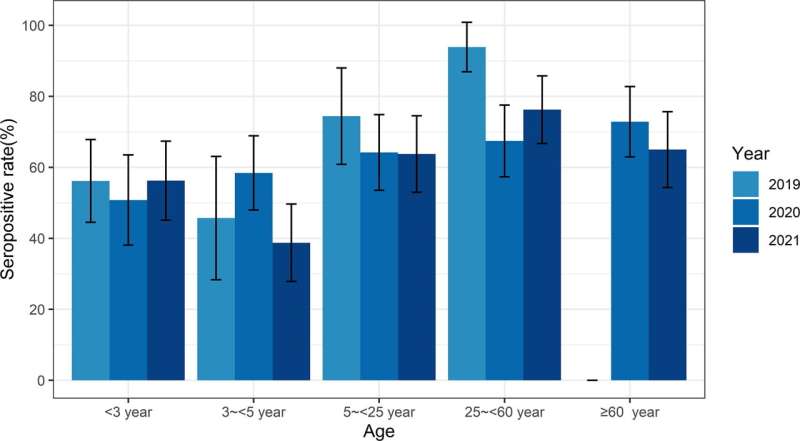
Microneutralization assay was performed on 1,000 representative serum samples. Notably, the seroprevalence and geometric mean titer (GMT) decreased significantly in 2020, and that in the below-three-year age group were increased and even higher than that in three- to five-year age group in 2019 and 2021, which was contrary to the authors' previous surveillance result and other studies in Guangzhou. Furthermore, a moderate decline of GMT level was observed following the vaccination, but the seropositive serums were still detected for 49 months after second immunization, suggesting the long-term persistence of the immunity.
This seroepidemiology study revealed relatively higher neutralizing antibody activity in the susceptible population after the EV-A71 vaccine was adopted in 2016 in Guangzhou. It may be one of the reasons for the lower epidemic activity of EV-A71 in Guangzhou from 2019 to 2021.
More information: Huimin Lian et al, Seroprevalence of human enterovirus A71 in Guangzhou, China, 2019–2021, Biosafety and Health (2023). DOI: 10.1016/j.bsheal.2023.05.002