This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
A new drug prevents epithelial-mesenchymal transition, metastasis and resistance to anti-cancer therapy

Metastases and resistance to chemotherapy are the main causes of treatment failure and mortality in cancer patients. Epithelial-mesenchymal transition (EMT), a process by which cancer cells detach from their neighboring cells and acquire invasive properties, plays a key role in the formation of metastases and the development of resistance to anti-cancer treatments. To date, there is no therapy targeting EMT in cancer.
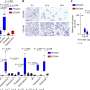
In a study published in Nature, researchers led by Pr Cédric Blanpain—WEL Research Institute investigator, director of the Stem Cells and Cancer Laboratory, Faculty of Medicine and professor at the Université libre de Bruxelles showed that netrin- 1, a molecule expressed by tumor cells in different types of cancers, stimulates the epithelial-mesenchymal transition (EMT) in tumor cells and a drug targeting netrin-1 blocks EMT in cancer.
Justine Lengrand, Ievgenia Pastushenko and Sebastiaan Vanuytven and her colleagues found that cancer cells presenting EMT express high levels of netrin-1 and its receptor UNC5B. Researchers have shown that increasing netrin-1 promotes EMT, while targeting netrin-1 decreases EMT.
In collaboration with NETRIS Pharma, which has developed a therapeutic antibody specifically blocking the interaction between netrin-1 and its receptor UNC5B, the ULB researchers have shown that the administration of the therapeutic antibody leads to a reduction in tumor formation but also blocks EMT in these tumors, which reduces their ability to give rise to metastases and sensitizes the tumor cells to chemotherapy.
"We are extremely happy and excited to have identified the first drug that can target EMT in vivo and therefore reduce the formation of metastases and resistance to chemotherapy," explains Justine Lengrand, the first author of the study.
After having demonstrated the effectiveness of the anti-netrin 1 antibody in preventing EMT in animal models, the ULB researchers then collaborated with researchers from the University of Lyon and Nétris Pharma to study the effect of this drug on EMT in patients with endometrial cancers.
The researchers and clinicians administered the anti-netrin antibody to patients in clinical trials in France. These studies showed that the administration of the therapeutic antibody was well tolerated and showed no toxicity. More importantly, they showed—on biopsies from the tumors taken before and after administration of the drug—that this therapy decreased EMT in patients with endometrial cancers.
"This is a major world premiere; we have discovered a new drug that can reduce EMT, decrease metastasis and stimulate the response to chemotherapy in preclinical models. In a second study, the researchers and clinicians provided the proof of principle for the medical application of our fundamental discovery and showed that the administration of the anti-netrin-1 antibody inhibits EMT in cancer patients," the researchers state.
"We have now to assess whether the administration of the anti-netrin-1 antibody and the reduction of the EMT will provide to the cancer patients a better clinical response to chemotherapy and immunotherapy," comments Professor Cédric Blanpain, the leader of the project.
The Belgo-French collaboration identifies novel innovative therapeutic combinations to sensitize tumors to chemotherapy and prevent tumor progression, the development of metastases and resistance to anti-cancer therapy.
"In the long term, it will be necessary to determine the effectiveness of this new therapy on the survival of patients with endometrial cancers and assess this efficacy of this new drug combination for the treatment of other types of cancers presenting EMT such as lung or breast cancers," comments Blanpain, the last author of the study.
More information: Patrick Mehlen, Netrin-1 blockade inhibits tumor growth and EMT features in endometrial cancer, Nature (2023). DOI: 10.1038/s41586-023-06367-z
Justine Lengrand et al, Pharmacological targeting of netrin-1 inhibits EMT in cancer, Nature (2023). DOI: 10.1038/s41586-023-06372-2