This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Epigenetically guided cancer therapy: Targeting H3K27me3 loss in pediatric brain tumors
A new editorial paper titled "Targeting H3K27me3 loss in pediatric brain tumors—a perspective on epigenetically guided cancer therapy" has been published in Oncotarget.
High-grade tumors of the central nervous system, including medulloblastoma, ependymoma and DMG (diffuse midline glioma, formerly known as DIPG, for diffuse intrinsic pontine glioma), constitute a major challenge in pediatric oncology. They are characterized by an aggressive growth and high relapse rates, and claim the lives of many pediatric cancer patients.
Both medulloblastoma and ependymoma are treated with surgical resection followed by adjuvant radiation therapy. DMG, on the other hand, diffusely infiltrates the brain stem making a resection virtually impossible. Thus, radiotherapy is the primary treatment modality for this tumor. While radiation temporarily attenuates the progression of DMG, this brain cancer remains incurable and most children succumb to their disease.
In his new editorial, Dr. Michael Goldstein from Johns Hopkins University School of Medicine discusses the extensively investigated molecular profiles of the aforementioned pediatric brain tumors demonstrating distinct epigenetic traits.
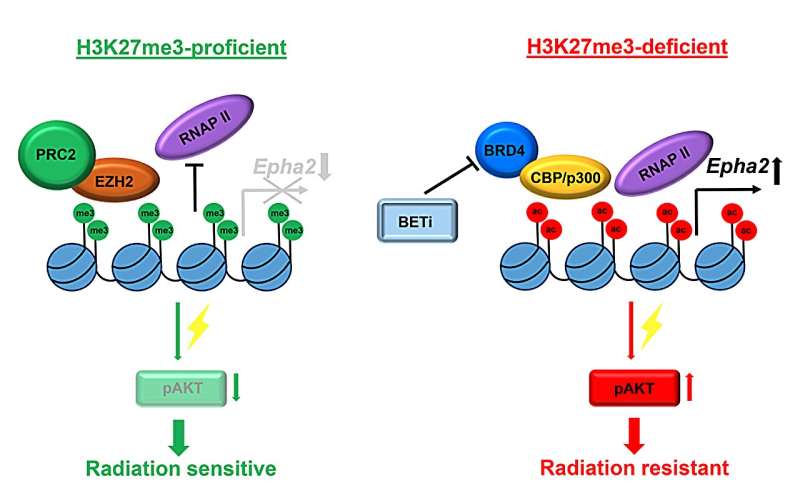
"Strikingly, a global loss of H3K27 tri-methylation (H3K27me3) as a result of the dominant-negative histone H3K27M mutation was found to be a hallmark of DMG occurring in the majority of the tumors," Dr. Goldstein states.
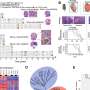
H3K27me3 is a product of the EZH2 histone methyltransferase affecting multiple cellular processes including transcription, chromatin structure and DNA damage response. Similarly, the aggressive PFA ependymoma subgroup is characterized by a lack of H3K27me3 due to an overexpression of the EZHIP protein that acts as an EZH2 inhibitor, whereas less aggressive PFB tumors retain normal H3K27me3 levels. However, no comprehensive analysis of H3K27me3 expression patterns in medulloblastoma has been performed, and the significance of this epigenetic mark in pediatric brain tumors has remained unknown.
Dr. Goldstein adds, "To address this, we have investigated the levels of the H3K27me3 histone mark and its role in treatment response of non-WNT/SHH medulloblastoma comprising group 3 and group 4 tumors. We demonstrated that about 50% of the tumors in patients with group 3 and group 4 medulloblastoma are H3K27me3 deficient. Strikingly, loss of H3K27me3 was associated with high relapse rates and poor survival."
More information: Michael Goldstein, Targeting H3K27me3 loss in pediatric brain tumors—a perspective on epigenetically guided cancer therapy, Oncotarget (2023). DOI: 10.18632/oncotarget.28427