This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Unveiling Australia's fastest next-gen mpox diagnostic tool
In a collaborative study published in The Lancet Microbe, a team of scientists, led by the Peter Doherty Institute for Infection and Immunity (Doherty Institute) and WEHI (Walter and Eliza Hall Institute of Medical Research), revealed MPXV-CRISPR—a powerful diagnostic tool capable of detecting monkeypox virus (MPXV) in clinical samples with acute precision and at a faster rate than any other method, thanks to the power of CRISPR technology. It is the first CRISPR-based diagnostic method in Australia specifically designed to target genetic sequences found only in MPXV.
While the CRISPR technology is most known for its genome editing capability, new applications have emerged, including leveraging it for the design of powerful and highly sensitive diagnostic tools.
The University of Melbourne's Dr. Soo Jen Low, a Research Officer at the Doherty Institute and co-first author of the study, said CRISPR-based diagnostic tools were like super-precise detectives that can quickly find specific clues (in this instance, genetic material) related to the presence of specific pathogens.
"To work, MPXV-CRISPR has to be 'programmed' to recognize the virus. We used a database of 523 MPXV genomes to carefully engineer 'guides' to bind to the specific part we are looking for on the viral DNA. Getting this right was crucial for the success of our diagnostic tool," said Dr. Low.
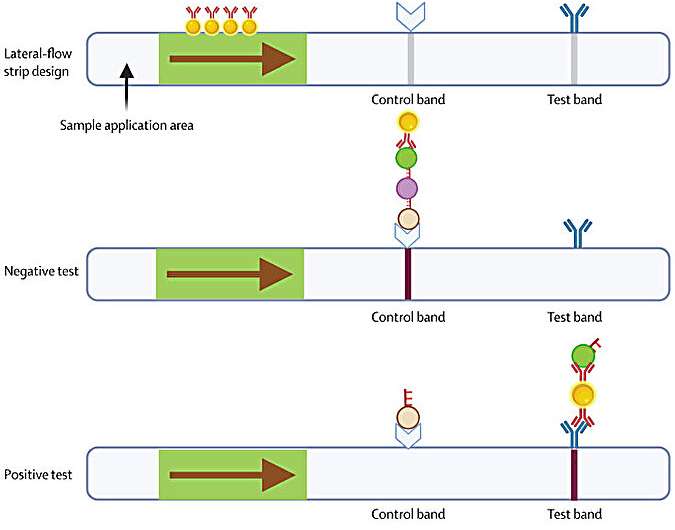
"In essence, when viral DNA is present in a clinical sample, the CRISPR system is guided to the target and subsequently emits a signal to indicate the presence of the virus. Our testing method can achieve sensitivity and precision levels comparable to the gold-standard PCR methods, but in a fraction of the time," said Dr. Low.
Matthew O'Neill, a Research Assistant at WEHI and co-first author of the paper, explained that the speed at which this new technology can provide a diagnosis is one of the groundbreaking features of MPXV-CRISPR.
"Currently, mpox diagnostics rely largely on centralized laboratory settings, where test results might not be available for up to several days after sample collection, depending on geographical and logistical considerations. In parallel, MPXV-CRISPR can detect the virus in just 45 minutes," said O'Neill.
In line with the WHO standards where diagnostic tests should be accurate, accessible and affordable, the team is working on adapting MPXV-CRISPR into a portable device, that could, one day, be deployed at points of care around the country for rapid, on-site detection of monkeypox virus.
Dr. Shivani Pasricha, a Senior Research Officer at WEHI, Junior Laboratory Head at the Doherty Institute, and co-senior author of the paper, said MPXV-CRISPR has the potential to revolutionize the way we manage mpox, making a meaningful impact on public health.
"By improving access to quick and reliable diagnoses around Australia, including in places with limited resources and in remote areas, this decentralized approach to testing could enable faster treatment and improve patient outcomes, while fast-tracking our capacity to manage future outbreaks," said Dr. Pasricha.
This research is the result of a collaboration between the Doherty Institute, WEHI, Melbourne Sexual Health Center and Monash University.
More information: Soo Jen Low et al, Rapid detection of monkeypox virus using a CRISPR-Cas12a mediated assay: a laboratory validation and evaluation study, The Lancet Microbe (2023). DOI: 10.1016/S2666-5247(23)00148-9