This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Exploring neurons' molecular highways: How a key protein regulates neuronal health
The Molecular Neuroscience Unit at the Okinawa Institute of Science and Technology (OIST) has made an important breakthrough by connecting sensory neurons' survival and pathology with the way messenger RNAs (mRNAs) are transported inside these cells.
This team of neurobiologists, comprised of Ph.D. student Sara Emad El-Agamy, Dr. Laurent Guillaud, and Prof. Marco Terenzio, has collaborated with Prof. Keiko Kono from OIST's Membranology Unit and Dr. Yibo Wu from the Riken Institute (currently at the University of Geneva). The project was led by Sara Emad El-Agamy, first author of the published study, as part of her Ph.D. work.
"Neurons have perhaps the most extreme morphology among cells, as they can vary in shape and extend over long distances in large mammals. For example, neurons that innervate a person's leg can be more than a meter long: they may have their nucleus near the spinal cord but sense a tickling sensation in the feet or pain in the big toe," explains Prof. Terenzio, who leads OIST's Molecular Neuroscience Unit.
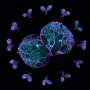
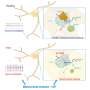
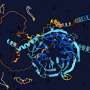
Neurons have long protrusions, called axons, within which molecules such as proteins, RNA, and organelles travel. This form of transport, from the center of the cell to the periphery and vice versa, is the cellular equivalent of a network of highways and trucks. The most important "trucks" responsible for propelling "cargoes" from the peripheral tips of neurons to their center, are part of a large complex of proteins called dynein. Malfunctions in this transport system can lead to several types of neuropathologies.
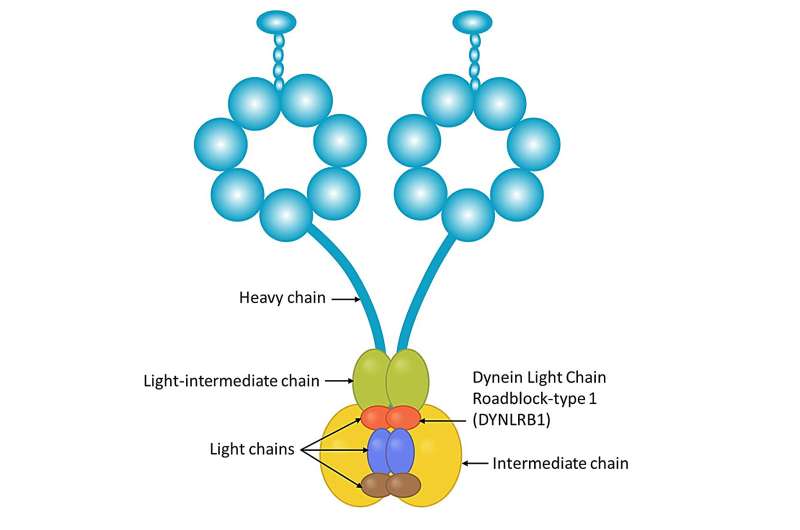
Dynein is a large and complex protein, composed of several subunits—or chains—which are classed by size. "We have studied a part of the dynein complex called Dynein Roadblock 1, or Dynlrb1 for short. In previous experiments, we demonstrated that this dynein subunit is clearly essential for the survival of neurons, but we had to figure out how it works," says Prof. Terenzio.
The researchers wanted to test an idea: if we think of dynein as trucks that move cargo inside the neurons, we could imagine that Dynlrb1 could affect the ability of the "dynein truck" to move or its ability to carry a cargo. To solve the riddle, the OIST team examined the proteins that interact with this dynein subunit.
Among the several interacting proteins, Sara Emad El-Agamy focused on the fragile X messenger ribonucleoprotein 1 (FMRP), which is well-known in the neurobiology field because it is related to a neurodevelopmental disorder (fragile X syndrome) and a neurodegenerative disease (fragile X-associated tremor/ataxia syndrome).
"Discovering that FMRP is part of the dynein cargo is particularly interesting. FMRP granules are composed of two types of molecules, proteins and messenger RNA (mRNA). mRNA are the template used by ribosomes to make proteins. Since I am really interested in the RNA biology of axons, I did not want to miss the opportunity to delve deeper into this topic," explains Prof. Terenzio.
Historically, axons were thought to lack RNA and protein synthesis machinery, with most of these processes believed to occur exclusively near the nuclei of neurons. However, relatively recent research has revealed that axons do indeed contain various RNA molecules. Since synthesizing proteins in the center of the cell and then shipping all these proteins to the tips of the neurons, like large cargo on trucks, would be a big investment of energy for the long neurons, neurons ship mRNA instead of proteins.
"A single mRNA can serve as a template to produce several proteins. By transporting mRNA instead of the final proteins, cells can conserve a substantial amount of energy, at least in theory," explains Prof. Terenzio.
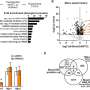
However, Sara also found out FMRP is trafficked from the periphery to the center. "These are typically believed to be trafficked from the center of the cell to the periphery. The fact that we showed that they are transported in the opposite direction was very surprising to us. This is a phenomenon that has just started to be described in the field and I believe it will be important in the future," says Prof. Terenzio.
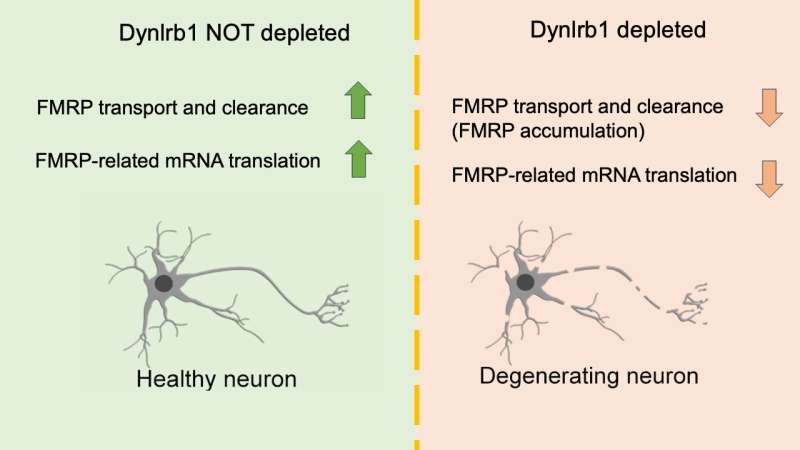
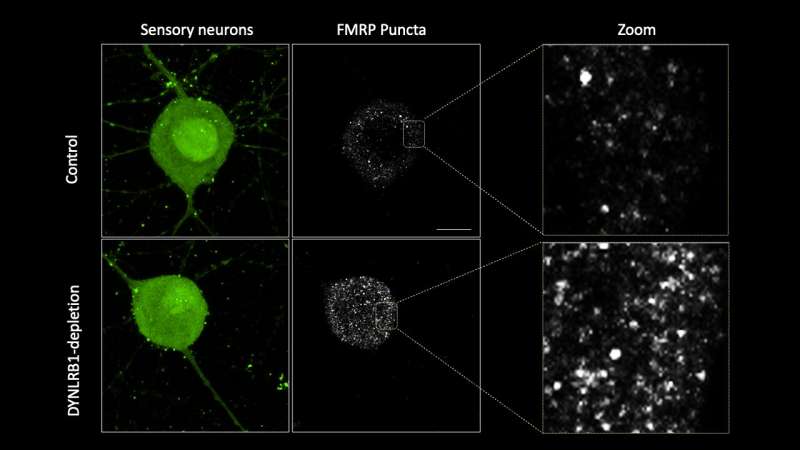
Finally, the team also discovered that the removal of Dynlrb1 causes FMRP to stall and accumulate in the cell bodies and axons of the sensory neurons. Since mRNA bound to FMRP gets trapped and cannot undergo translation into proteins, the researchers hypothesize that Dynlrb1 plays a vital role in neuronal health. In other words, damages to Dynlrb1 could impede the production of essential proteins, jeopardizing neuron survival.
"Our next research question is to understand which proteins cannot be produced when Dynlrb1 is malfunctioning or absent. The data we acquired will help in understanding what supports neuronal survival and as an extension neuronal death. This could be leveraged to find new therapeutic approaches for neurodegenerative diseases," Prof. Terenzio concludes.
The findings are published in the journal Molecular & Cellular Proteomics.
More information: Sara Emad El-Agamy et al, FMRP Long-Range Transport and Degradation Are Mediated by Dynlrb1 in Sensory Neurons, Molecular & Cellular Proteomics (2023). DOI: 10.1016/j.mcpro.2023.100653