This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Hospital superbugs: Could one vaccine rule them all?

What if a vaccine, given to patients just before or after arriving at the hospital, could protect them against lethal superbugs that lurk in health care settings?
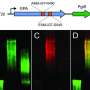
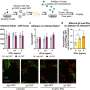
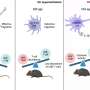
That's the premise behind an experimental vaccine invented by a USC-led team and patented by the university. Researchers designed the formula to prevent serious infections from drug-resistant pathogens. A new study shows that a single dose, administered in mouse models, put immune cells into "Incredible Hulk" mode, providing rapid protection against eight different bacteria and fungi species.
"It's an early warning system. It's like Homeland Security putting out a terror alert. 'Everybody, keep your eyes open. Keep an eye out for suspicious packages,'" said senior author Brad Spellberg, chief medical officer at the USC-affiliated Los Angeles General Medical Center (formerly LAC+USC).
The study appears October 4 in Science Translational Medicine.
The USC Stevens Center for Innovation, the technology licensing office for USC, successfully filed one patent for the vaccine and is pursuing others.
"The pandemic stimulated unprecedented innovation in vaccine development, where federal funding and university-industry partnerships were game changers for translating promising discoveries from academic labs for the good of all," said Ishwar K. Puri, senior vice president of research and innovation at USC.
"We are both pleased and proud of the critical support the USC Stevens Center provided to enable the development of ExBaq's experimental vaccine that protects vulnerable populations from serious infections."
Every year, health care acquired infections kill more than 90,000 people in the United States and rack up health care costs between $28 billion and $45 billion. On any given day, about one in 31 hospital patients has at least one such infection, according to the Centers for Disease Control and Prevention.
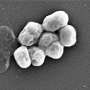
In many cases, infections are caused by so-called superbugs such as MRSA—short for methicillin-resistant Staphylococcus aureas —or Acinetobacter baumannii. The infections spread via contaminated surfaces or equipment, such as catheters or ventilators, or though person-to-person spread, often from contaminated hands. Risk is highest among ICU patients who may suffer surgical site infections, bloodstream infections, urinary tract infections and ventilator-associated pneumonia.
Typical vaccines usually prompt the body to make antibodies against a specific pathogen. Despite the high incidence of health care-acquired infections, there are currently no FDA-approved vaccines that prevent the most serious, antibiotic-resistant infections.
"Even if there were such vaccines, multiple vaccines would have to be deployed simultaneously to protect against the full slate of antibiotic-resistant microbes that cause health care-acquired infections," said Brian Luna, Assistant Professor of Molecular Microbiology and Immunology at Keck School of Medicine of USC.
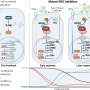
The experimental vaccine takes an entirely different approach: It gooses the body's preexisting supply of pathogen-gobbling immune cells called macrophages, which engulf and digest bacteria, fungi and other bad actors. These activated fighters, found in all tissues, quickly neutralize incoming invaders which might otherwise multiply rapidly and overwhelm the body's defenses.
"This is very different from developing new antibiotics," said Jun Yan, a Ph.D. student at Keck School of Medicine of USC and the study's first author. "This is using our own immune system to fight against different superbugs, which is a different approach than everybody else."
The vaccine is comprised of just three ingredients, two of which are already used in FDA-approved vaccines. A third component is a tiny piece from the surface of a fungus commonly found on human skin.
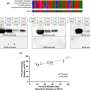
Tested in two independent labs, the vaccine works within 24 hours and lasts for up to 28 days. In lab models, the number of pathogen-eating immune cells in the blood increased dramatically, and survival time of invasive blood and lung infections improved. Early data suggest that a second dose could extend the window to prevent infection.
To develop the vaccine, Spellberg, Luna, Yan and Travis Nielsen, who earned his Ph.D. in the Keck School of Medicine's biomedical and biological sciences program before going to medical school, formed the startup ExBaq LLC.
ExBaq's founders have begun talking with potential pharmaceutical partners who might be interested in further developing the vaccine for human clinical trials.
The next step is getting guidance from the FDA on the requirements to complete preclinical studies and submit and Investigational New Drug Application (IND) in 2024. The first such trial would be done in healthy volunteers to find the right dose of vaccine that is safe and triggers the same kind of immune response in people as seen in the mice.
In addition to Yan, Luna, Spellberg and Nielsen, other authors of the paper are Peggy Lu, Yuli Talyansky, Matt Slarve and Hernan Reza, all of the Department of Molecular Microbiology and Immunology at the Keck School of Medicine of USC; Boris Novakovic of University of Melbourne; Mihai Netea of Radboud University; and Ashley Keller, Troy Warren, Antonio DiGiandomenico and Bret Sellman, all of AstraZeneca.
More information: Jun Yan et al, A Protein-Free Vaccine Stimulates Innate Immunity and Protects against Nosocomial Pathogens, Science Translational Medicine (2023). DOI: 10.1126/scitranslmed.adf9556. www.science.org/doi/10.1126/scitranslmed.adf9556