November 24, 2023 report
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Reactivating silenced fetal hemoglobin genes could counter sickle cell–related diseases
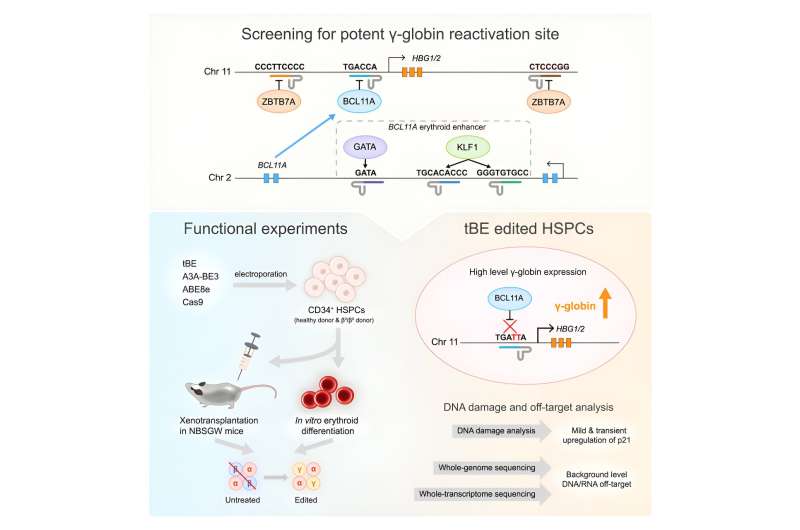
Researchers from multiple institutions in China have found a way to use gene editing to reactivate dormant fetal oxygen-transporting proteins in adult blood cells to potentially reverse a wide range of blood disorders.
In a paper, "Base editing of the HBG promoter induces potent fetal hemoglobin expression with no detectable off-target mutations in human HSCs," published in Cell Stem Cell, the team compares gene editing techniques while formulating a method that could have important clinical applications.
Fetal gamma (γ) globin is normally replaced by adult (β) hemoglobin during development. In an odd quirk of evolution, only humans and a few types of monkeys are known to switch from γ to β gene expression.
The genes producing the fetal hemoglobin become silenced and dormant after the genetic switch by repressors such as BCL11A and ZBTB7A, whose binding motifs have been identified as targets for reactivation.
β-hemoglobinopathies, including β-thalassemia and sickle cell disease, result from mutations in the HBB gene, leading to impaired β-globin production and resulting in anemia, impaired oxygen delivery to tissues and possible multi-organ tissue damage.
The researchers experimentally discovered that reactivating γ-globin expression could be developed into a universal therapeutic strategy for these conditions.
Six regulatory motifs (BCL11A enhancer and HBG1/2 promoter regions) were targeted using a recently developed cytosine base editor—transformer base editor (tBE). The team compared tBE with other base editors and Cas9 nuclease for efficiency and off-target effects.
In the study, tBE exhibited comparable or higher editing efficiency than other editors across the targeted motifs. Comprehensive analysis revealed no detectable off-target mutations in tBE-edited cells, indicating the potential of tBE as a safer and more potent treatment strategy for β-hemoglobinopathies.
Experiments conducted with patient-derived cells highlighted that disrupting the BCL11A binding sites within the HBG1/2 promoters led to the highest levels of γ-globin expression. Xenotransplantation in mice showed persistent editing in HSCs and their progenies, maintaining engraftment potential and differentiation ability.
The increased γ-globin expression observed due to tBE-mediated editing signifies a promising therapeutic avenue for β-hemoglobinopathies.
While editing methods and not direct clinical outcomes were the focus of the study, the substantial enhancement in γ-globin expression levels strongly suggests potential clinical benefits, including symptom alleviation and improved disease management for individuals affected by β-hemoglobinopathies.
More information: Wenyan Han et al, Base editing of the HBG promoter induces potent fetal hemoglobin expression with no detectable off-target mutations in human HSCs, Cell Stem Cell (2023). DOI: 10.1016/j.stem.2023.10.007
© 2023 Science X Network