This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Two COVID-19 vaccines show robust boosting potential in clinical trial
Two Melbourne-made COVID-19 vaccines have shown strong potential to be an improved approach for boosting immunity to SARS-CoV-2 variants according to interim results of a Phase 1 clinical trial.
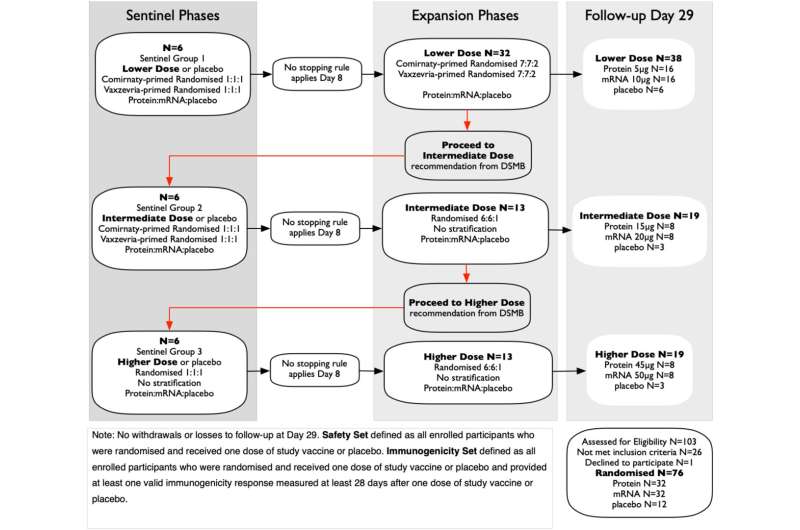
Published in eBioMedicine, 76 healthy adults from Melbourne, aged 18 to 64 who were previously vaccinated with licensed SARS-CoV-2 vaccines, were randomized to receive a fourth dose of a novel protein vaccine, mRNA vaccine or placebo.
Both vaccines demonstrated strong boosting capabilities in a highly immune population and a remarkable breadth of immune response, including against omicron sub-variants. In addition, no safety signals were observed with either candidate.
The two vaccine candidates, created by researchers at the Peter Doherty Institute for Infection and Immunity (Doherty Institute) and the Monash Institute of Pharmaceutical Sciences (MIPS), are distinct from most existing vaccines that are in use around the globe because they focus the immune response on the tip of the SARS-CoV-2 spike protein, called the receptor binding domain (RBD). The RBD enables the virus to enter and infect cells in the body and elicits over 90 percent of neutralizing antibodies (antibodies that can block the virus) following SARS-CoV-2 infection.
The two candidates are:
- RBD protein vaccine—uses an engineered part of the virus protein, rather than genetic material or another virus, to elicit an immune response.
- RBD mRNA vaccine—represents the virus genetic sequence of mRNA that codes for the tip of the spike, which leads to production of the RBD protein in the recipient.
University of Melbourne Professor Terry Nolan, Head of the Vaccine and Immunization Research Group at the Doherty Institute, which led the Phase 1 first-in-human trial, said the team was exceptionally pleased by the results.
"Post-market studies of omicron-directed, whole spike bivalent mRNA booster vaccines have shown modest increases in immune responses to omicron variants compared to ancestral vaccine boosts," said Professor Nolan.
"Because our two vaccines focus the immune response on the receptor binding domain, they avoid unhelpful immune responses against other parts of the spike protein and could therefore provide a more efficient approach for boosting immunity to the virus, presenting a strong case to proceed to Phase 2 clinical trials."
Professor Colin Pouton of MIPS, who led the development of the RBD mRNA vaccine, said the mRNA vaccine showed a strong immune response, even at the lowest dose tested.
"So far, both preclinical and clinical studies have shown our RBD mRNA vaccine to provide a strong boost at low doses, suggesting the very real potential to develop a multivalent vaccine, on an annual basis and to protect against emerging new variants of COVID-19, which are believed to be the root cause behind the ongoing 'waves' we are still experiencing," said Professor Pouton.
"New strategies are still needed to improve efficacy of COVID-19 variant vaccines and to reduce death rates, particularly among older and vulnerable patients. In the case of our mRNA vaccine, we've also seen early potential to address the issue of immune imprinting, which will also need to be a critical feature for a next-generation vaccine."
The team is now exploring options for progressing these vaccines to Phase 2 trials.
More information: Terry M. Nolan et al, Interim results from a phase I randomized, placebo-controlled trial of novel SARS-CoV-2 beta variant receptor-binding domain recombinant protein and mRNA vaccines as a 4th dose booster, eBioMedicine (2023). DOI: 10.1016/j.ebiom.2023.104878