This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
YTHDF2's role in promoting acute myeloid leukemia: The key lies in microRNA processing
In a study published in the journal Genes & Diseases, researchers from Beckman Research Institute of City of Hope, explored the impact of m6A modification on the maturation process of precursor microRNAs (pre-miRNAs) and its potential role in tumorigenesis. They have uncovered a novel mechanism behind the progression of acute myeloid leukemia (AML), a life-threatening blood cancer characterized by rapid expansion of abnormally differentiated hematopoietic cells.
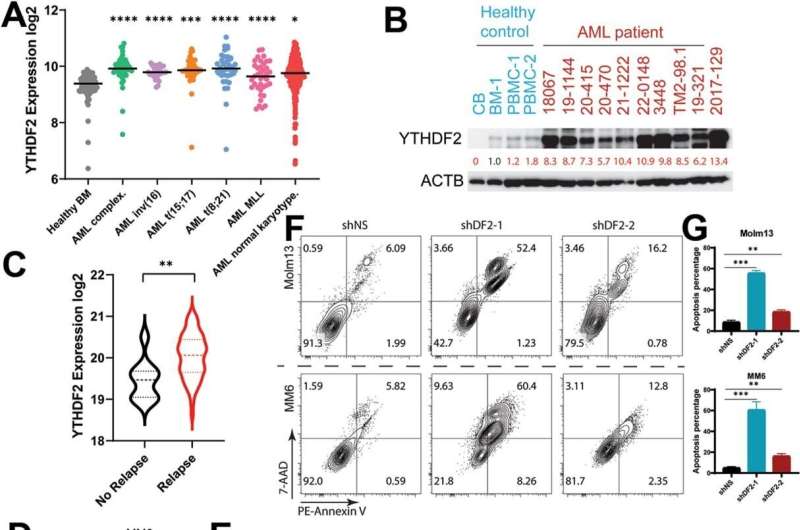
Despite advances in treatment over the years, the five-year survival rate for AML patients stands at a mere 30%, underscoring the dire need for breakthroughs in understanding and treating this disease. At the heart of this discovery is the RNA m6A reader, YTHDF2. This protein, found in elevated levels in AML patients—particularly those who have relapsed, plays a significant role in the disease's progression.
The findings of the research indicate that YTHDF2 accelerates AML by enhancing the expression of a particular microRNA, miR-126-3p (or miR-126). This investigation was particularly centered on the RNA m6A reader YTHDF2 and its overexpression in acute myeloid leukemia (AML) patients.
In a significant breakthrough concerning AML, especially in patients who have relapsed, researchers observed a pronounced overexpression of YTHDF2, highlighting its potential oncogenic role. This protein not only amplifies the expression of miR-126-3p, commonly recognized as miR-126, by assisting the maturation of its precursor form, pre-miR-126, but also astutely identifies the m6A modification within it. By partnering with AGO2, a pivotal regulator, YTHDF2 facilitates the progression of AML.
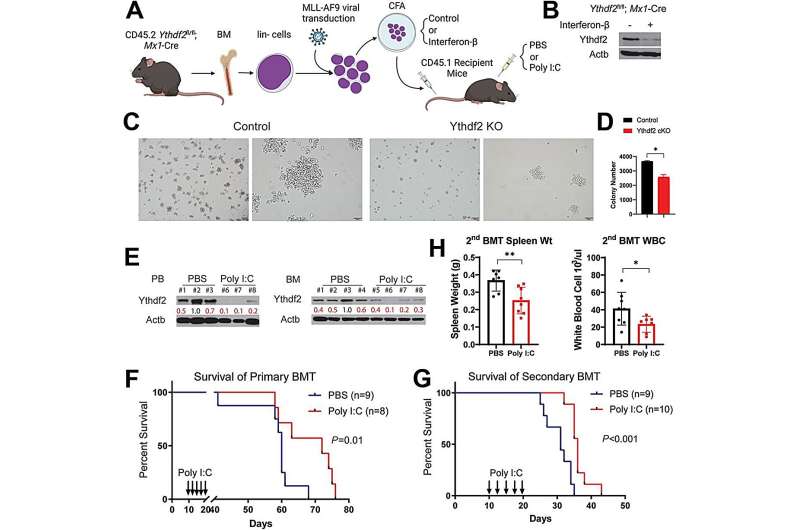
Clinically, a direct correlation between YTHDF2's levels and miR-126 has been established, while its relation with miR-126's target genes is inversely proportional. This discovery accentuates miR-126's cardinal role in the YTHDF2 mechanism, where enhancing its expression can potentially offset the impacts of YTHDF2 depletion. This groundbreaking study uncovers a previously unexplored YTHDF2/miR-126 axis in AML progression. By understanding this mechanism, we identify the promising therapeutic potential of targeting this axis for AML treatment. The findings also emphasize m6A's influential role in pre-miRNA processing that may contribute to cancer development.
Understanding the intricate roles of m6A, YTHDF2, and miR-126 in AML progression offers promising avenues for developing targeted treatments. With miRNAs like miR-126 playing a critical role in AML and other diseases, insights from this study can pave the way for therapies not only for AML but potentially for other conditions impacted by miRNA dysregulation.
More information: Zheng Zhang et al, RNA m6A reader YTHDF2 facilitates precursor miR-126 maturation to promote acute myeloid leukemia progression, Genes & Diseases (2023). DOI: 10.1016/j.gendis.2023.01.016