This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers examine the benefits of COVID-19 nasal spray vaccination
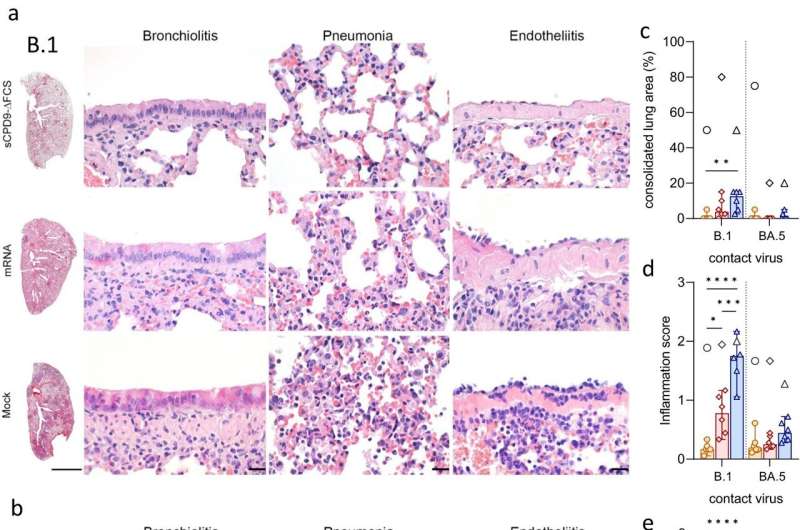
A research team at Freie Universität Berlin has conducted a comprehensive comparison of the efficacy of a mucosal, replication-competent but fully attenuated virus vaccine, sCPD9-ΔFCS, with the monovalent mRNA vaccine BNT162b2 in preventing the transmission of SARS-CoV-2 variants.
The study, titled "An Intranasal Live-Attenuated SARS-CoV-2 Vaccine Limits Virus Transmission," is published in Nature Communications. The research addresses a critical challenge facing current vaccination strategies—namely, the limited ability of intramuscularly administered vaccines to induce robust mucosal immune responses in the upper respiratory tract, the primary site of infection and virus shedding.
The researchers compared the efficacy of the live attenuated vaccine (LAV) sCPD9-ΔFCS and a monovalent mRNA vaccine in preventing the spread of two SARS-CoV-2 variants: the ancestral B.1 and the omicron BA.5 in Syrian hamsters. They investigated the performance of the vaccines in two different scenarios.
In the first scenario, they evaluated the protective efficacy of the two vaccines by exposing vaccinated Syrian hamsters to infected antagonists. Here, the LAV vaccine completely blocked infection, while the mRNA vaccine offered no significant protection against infection.
In the second scenario, the transmission of the challenge virus from vaccinated and subsequently infected hamsters to unsuspecting contacts was investigated. In this scenario, transmission was blocked or strongly suppressed by the LAV vaccine but not by the mRNA vaccine. These results clearly showed that the LAV sCPD9-ΔFCS significantly outperformed the mRNA vaccine in preventing virus transmission in both scenarios.
Freie Universität Berlin virologist Dr. Jakob Trimpert, one of the lead authors of the study, said, "Our results provide compelling evidence for the benefits of locally administered live attenuated vaccines over intramuscularly administered mRNA vaccines. This is a significant advance in improving our ability to prevent infection and reduce virus transmission, particularly in the context of emerging SARS-CoV-2 variants."
According to the team, the COVID-19 pandemic has posed unprecedented challenges to research and society, and the continued spread of SARS-CoV-2 underscores the need for innovative vaccination strategies. The LAV sCPD9-ΔFCS, which was developed by de-optimizing codon pairs, offers a promising solution as it induces strong mucosal immunity in the respiratory tract and systemic immunity against a range of SARS-CoV-2 antigens.
"Our study has addressed the shortcomings of existing vaccines and underscored the potential of intranasally administered vaccines to create a more effective barrier against infection, prevent virus replication and attenuate transmission," said Dr. Dusan Kunec, research group leader at the Institute of Virology at Freie Universität Berlin, co-author and lead researcher of the study.
Rocketvax founders Dr. Vladimir and Dr. Natasa Cmiljanovic said, "The impressive preclinical results consistently confirm the advantages of our lead SARS-CoV-2 live vaccine candidate sCPD9-ΔFCS, administered intranasally, over intramuscularly administered mRNA vaccines, prompting us to accelerate its further development and translation into clinical settings for its further validation."
According to the research team, the results of the study have significant implications for the future of COVID-19 vaccination strategies. The superiority of sCPD9-ΔFCS in preventing transmission points to a promising avenue for further research and development of intranasal vaccines that offer a potential solution to the challenges posed by emerging variants.
This study provides compelling evidence for the superiority of the intranasally administered live attenuated vaccine sCPD9-ΔFCS in preventing SARS-CoV-2 transmission. The study underscores the importance of exploring alternative vaccination approaches to address the evolving landscape of the COVID-19 pandemic.
More information: Julia M. Adler et al, An intranasal live-attenuated SARS-CoV-2 vaccine limits virus transmission, Nature Communications (2024). DOI: 10.1038/s41467-024-45348-2




















