This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers identify critical pathway responsible for melanoma drug resistance
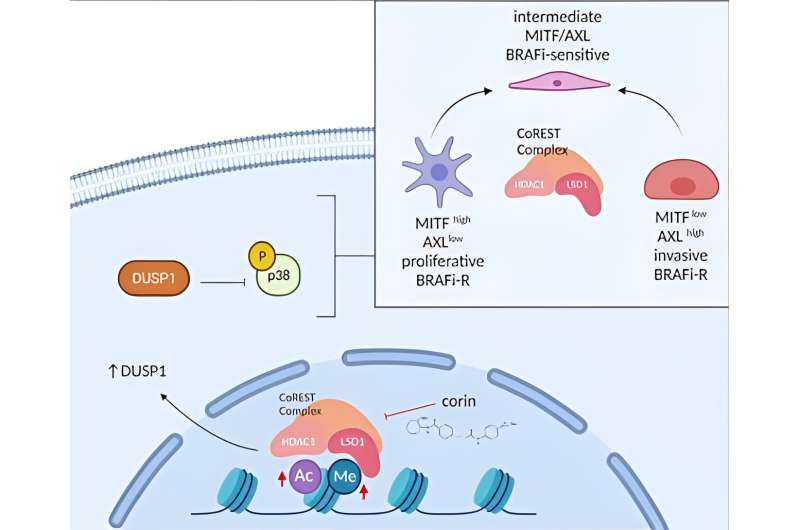
One of the major challenges in cancer research and clinical care is understanding the molecular basis for therapeutic resistance as a major cause of long-term treatment failures. In cases of melanoma, the main targeted therapeutic strategy is directed against the mitogen-activated protein kinase (MAPK) pathway. Unfortunately, in the vast majority of these patients, resistance to MAPK inhibitor therapies develops within one year of treatment.
In a new study from Boston University Chobanian & Avedisian School of Medicine, researchers have identified a specific epigenetic pathway involved in the development of therapy resistance in melanoma and have identified a promising chemical reagent that can successfully resensitize therapy-resistant cancers to targeted therapies.
The work is published in the Journal of Clinical Investigation.
"Although the cancer research community has been very successful in developing specific targeted therapies for the genetic events driving many cancers, most patients are unable to be cured of their cancers due to acquired resistance mechanisms. We are excited about the broader implications this study has for the potential treatment of patients with acquired resistance to cancer therapies," said co-corresponding author Rhoda Alani, MD, the Herbert Mescon Chair of Dermatology at the school.
In collaboration with investigators from Brigham and Women's Hospital and Harvard Medical School, the researchers used both cancer cell lines grown in the lab and human melanoma tumors grown in experimental models to evaluate the epigenetic influences associated with melanoma progression, and their response to targeted therapies.
They determined that a significant resistance mechanism for melanoma involves the epigenetic mediator, CoREST, and that this treatment resistance can be targeted by corin, a recently designed dual-functioning small molecule inhibitor of CoREST. The study provides the molecular rationale for how CoREST complex targeting can be harnessed for an important therapeutic goal in a broad range of epigenetic contexts, including melanoma.
Alani expects the clinical implications for this research to be significant as it is likely that similar epigenetic events are associated with treatment resistance in other cancers, and that the development of novel high-specificity epigenetic inhibitors like corin may allow for the resensitization of these tumors to effective targeted therapies and possible disease remission.
The researchers hope this study will lead to the development of more specific targeted epigenetic therapies for cancer and other diseases with minimal side effects.
"We also expect that these studies will result in the improved efficacy of anticancer targeted drugs while limiting the development of drug resistance in cancers through the combined use of targeted epigenetic therapies," added Alani, who also is chief of dermatology at Boston Medical Center.
In addition to Alani, the other corresponding author is Philip Cole, MD, Ph.D., (Brigham and Women's Hospital and Harvard Medical School).
More information: Muzhou Wu et al, The CoREST Repressor Complex Mediates Phenotype Switching and Therapy Resistance in Melanoma, Journal of Clinical Investigation (2024). DOI: 10.1172/JCI171063


















