This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Q&A: Supercomputer simulations find new starting point for HIV therapies
Molecular simulations on the JUWELS supercomputer show how the HIV-1 virus is able to evade the human immune system. The newly discovered mechanism could prove to be the Achilles' heel of the virus and open new doors for HIV therapies.
HIV infections and AIDS are among the most common and threatening infectious diseases worldwide. Highly effective therapies are now available. However, a complete cure or vaccination that protects against infection is not yet possible.
Researchers now believe that the virus has jumped from chimpanzees and gorillas to humans several times. Using molecular dynamics simulations on the JUWELS supercomputer at Forschungszentrum Jülich, Prof. Holger Gohlke from Forschungszentrum Jülich and Heinrich Heine University Düsseldorf and Prof. Carsten Münk from the University Hospital of Düsseldorf have now been able to understand in detail for the first time a step through which the virus has adapted to human cells. Their study is published in the Proceedings of the National Academy of Sciences.
Prof. Gohlke, what was the motivation for this study?
HIV-1 viruses are the evolutionary result of rare successful transmissions of the simian immunodeficiency virus (SIV) to humans. Humans are immune to SIV. We therefore wondered how the pandemic group HIV-M is able to evade the human immune system. There are also other, nonpandemic strains (HIV-N, HIV-O and HIV-P groups), which are very rare and have only been found in a few people infected in West Africa.
Vertebrates use several cellular restriction factors to prevent the replication, i.e., multiplication, of HIV. However, it has so far remained unclear whether the low incidence of non-M HIVs is related to the activity of these antiviral factors, i.e., whether they limit the spread of the virus among humans. There is a particular focus here on TRIM5α, a protein that is found in the cells of most primates and is effective against various infections and retroviruses.
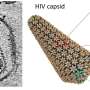
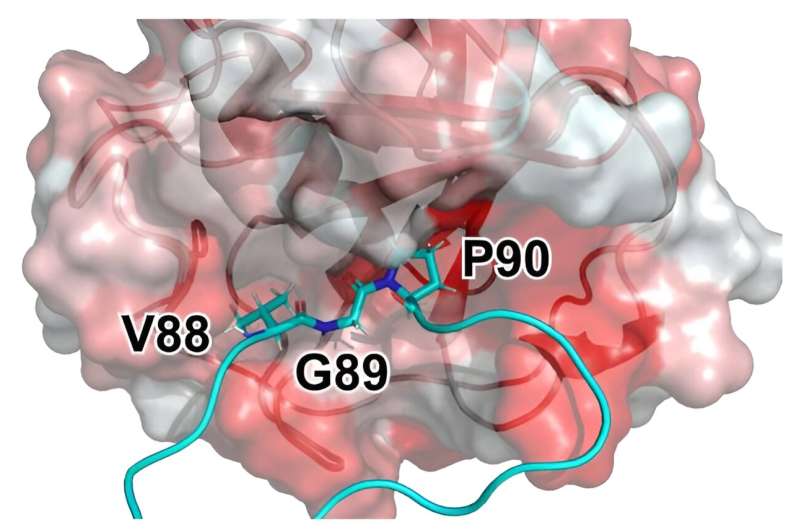
TRIM5α binds to the capsid, the shell that encloses the viral genome. The activity of this protein is partly dependent on cyclophilin A (CYPA), another protein which has catalytic properties, i.e., it is able to accelerate certain reactions. One of the main objectives of this work was to understand the mechanism.
What particular challenges were associated with the molecular dynamics simulations?
To start the simulations, a structural model of the HIV-1 M capsid first had to be produced with CYPA, which was then used to generate the other complexes for HIV-N, HIV-O, and HIV-P as well as other variants through "mutations in the computer." To assess the influence of CYPA on TRIM5, biased MD simulations were performed to simulate the conversion catalyzed by CYPA in the capsid. In addition, the conversion in the capsid without CYPA was simulated for comparison.
A total of 16 systems were simulated, with 121 individual steps for each system. This resulted in a high number of independent simulations that had to be performed. The simulations also had to be extensive in order to obtain highly precise results. This was necessary to gain detailed insights into the subtle differences between the various HIV groups and the role of CYPA.
What is the potential of the weak spot you have discovered in terms of the treatment of HIV?
The results of our simulations indicate that the speed of the reaction catalyzed by CYPA may play a crucial role in the protection of HIV-1 M and, therefore, in its pandemic property. Our data suggest that CYPA subtly alters the capsid—the shell that encloses the viral genome.
These induced changes appear to have a severe impact on the viral infection. It is the binding of CYPA which likely ensures that the human HIV-1 M virus is protected from the antiviral protein TRIM5α. The study has therefore identified a new "weak spot" of HIV-1 and could lead to new HIV-1 drugs that suppress the binding of CYPA to the virus in the cell or modulate the activity of CYPA.
More information: Augustin P. Twizerimana et al, The cyclophilin A-binding loop of the capsid regulates the human TRIM5α sensitivity of nonpandemic HIV-1, Proceedings of the National Academy of Sciences (2023). DOI: 10.1073/pnas.2306374120