This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
reputable news agency
proofread
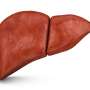
Study shows seladelpar beneficial for patients with primary biliary cholangitis
For patients with primary biliary cholangitis, more receiving the peroxisome proliferator-activated receptor delta (PPARδ) agonist seladelpar have a biochemical response and alkaline phosphatase normalization, according to a study published online Feb. 21 in the New England Journal of Medicine.
Gideon M. Hirschfield, Ph.D., from the University Health Network in Toronto, and colleagues conducted a phase 3, 12-month, double-blind placebo-controlled trial involving patients with primary biliary cholangitis with inadequate response to or who had a history of unacceptable side effects with ursodeoxycholic acid. One hundred ninety-three patients were randomly assigned to receive oral seladelpar at a dose of 10 mg daily or placebo in a 2:1 ratio. The primary end point was a biochemical response.
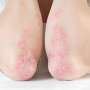
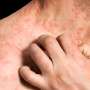
The researchers found that the percentage of patients with a biochemical response was higher in the seladelpar group versus the placebo group (61.7 versus 20.0%). In addition, significantly more patients who received seladelpar had normalization of the alkaline phosphatase level (25.0 versus 0%). Compared with placebo, seladelpar resulted in a greater reduction in the score on the pruritus numerical rating scale (least-squares mean change from baseline, −3.2 versus −1.7). Overall, 86.7 and 84.6% of patients in the seladelpar and placebo groups, respectively, had adverse events; for serious adverse events, the corresponding values were 7.0 and 6.2%.
"The selective PPARδ agonist seladelpar elicited biochemical responses while also reducing pruritus in patients with primary biliary cholangitis who had had an inadequate response to or who had a history of unacceptable side effects with ursodeoxycholic acid," the authors write.
More information: Gideon M. Hirschfield et al, A Phase 3 Trial of Seladelpar in Primary Biliary Cholangitis, New England Journal of Medicine (2024). DOI: 10.1056/NEJMoa2312100
David N. Assis, Advancing Second-Line Treatment for Primary Biliary Cholangitis, New England Journal of Medicine (2024). DOI: 10.1056/NEJMe2312745
Bernd Schnabl et al, PPAR Agonists in Primary Biliary Cholangitis, New England Journal of Medicine (2024). DOI: 10.1056/NEJMe2313802
Copyright © 2024 HealthDay. All rights reserved.