This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Experimental gene therapy for giant axonal neuropathy shows promise in clinical trial
An investigational gene therapy for a rare neurodegenerative disease that begins in early childhood, known as giant axonal neuropathy (GAN), was well tolerated and showed signs of therapeutic benefit in a clinical trial led by the National Institutes of Health (NIH).
Currently, there is no treatment for GAN and the disease is usually fatal by 30 years of age. Fourteen children with GAN, ages 6 to 14 years, were treated with gene transfer therapy at the NIH Clinical Center and then followed for about six years to assess safety. Results of the early-stage clinical trial are published in the New England Journal of Medicine.
The gene therapy uses a modified virus to deliver functional copies of the defective GAN gene to nerve cells in the body. It is the first time a gene therapy has been administered directly into the spinal fluid, allowing it to target the motor and sensory neurons affected in GAN. At some dose levels, the treatment appeared to slow the rate of motor function decline.
The findings also suggest regeneration of sensory nerves may be possible in some patients. The trial results are an early indication that the therapy may have favorable safety and tolerability and could help people with the rapidly progressive disease.
"One striking finding in the study was that the sensory nerves, which are affected earliest in GAN, started 'waking up' again in some of the patients," said Carsten G. Bonnemann, M.D., senior author and chief of the Neuromuscular and Neurogenetic Disorders of Childhood Section at the National Institute of Neurological Disorders and Stroke (NINDS), part of NIH. "I think it marks the first time it has been shown that a sensory nerve affected in a genetic degenerative disease can actually be rescued with a gene therapy such as this."
Participants in this "first-in-human" trial, which began in 2015, received a single dose of the gene therapy, called scAAV9/JeT-GAN, through an injection into the fluid surrounding the spine. The first two patients received the lowest dose of the gene transfer, which was increased in subsequent patients.
Four dose levels were tested over the course of the trial, which were estimated based on results from studies in animal models. Only one serious adverse event—a fever—was potentially linked to the gene therapy.
The treatment resulted in 129 related adverse events of lesser seriousness, including headache, back pain, irregular heart rhythms, and inflammation in spinal fluid that was treated with corticosteroids. Two patients who were older and received the lowest-dose therapy died during the study period due to events related to their underlying disease.
In addition to safety, Dr. Bonnemann and his colleagues also assessed motor function scores and tests of nerve function among the study participants. With increasing dose levels, they found the probability of any slowing of motor decline was 44%, 92%, 99%, and 90%, respectively. As GAN progresses, electrical measures of sensory nerves decline and eventually disappear. With gene therapy, 6 of 14 patients regained sensory nerve response after treatment—electrical measures increased, stopped declining, or became measurable after being absent.
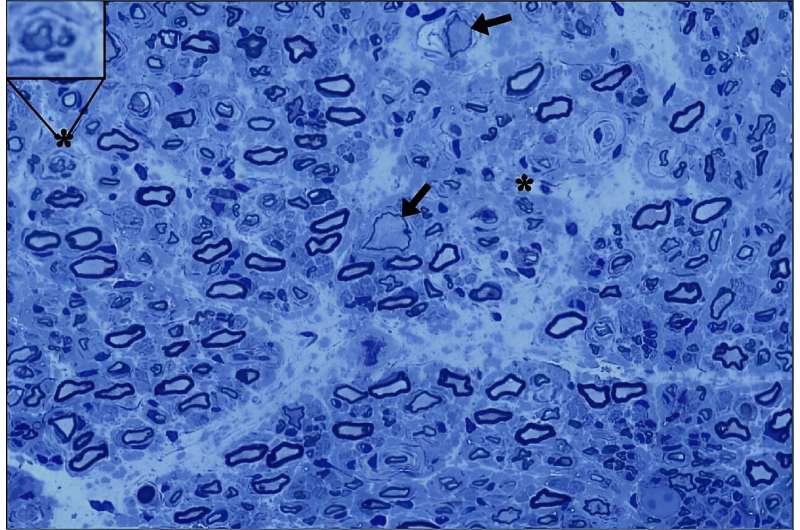
Mutations to the GAN gene result in an inability to break down intermediate filaments, which are cellular structures that make up the framework of nerve cell extensions called axons. Axons are essential for transmission of signals between brain cells. The disease name refers to the enlarged and bloated appearance of the axon under the microscope. As GAN progresses, the axons of motor and sensory nerves break down, resulting in difficulty with movement and sensation because nerve cells cannot communicate with each other.
The first symptoms of GAN are often a clumsy and unsteady gait, becoming evident as early as 2 or 3 years of age. The disease progresses so that by age 8 or 9, patients typically require the use of a wheelchair, followed by increasingly limited use of the arms and little to no use of their legs. In the later stages, people with GAN often require breathing assistance and a feeding tube.
This trial could also benefit gene therapy for other diseases. Researchers testing other gene therapies have already adopted direct administration into the spinal fluid, which requires lower doses compared to usual delivery into the bloodstream by vein.
Injecting into the spinal fluid also reduces the likelihood of an immune response, which enables patients who have developed immunity to adeno-associated virus (AAV), the common virus used as the gene delivery system in the therapy, to potentially receive treatment. Previously, children carrying antibodies to AAV from natural exposure to the virus would have been excluded from gene therapy because of their immune reaction.
Scientists will continue evaluating the scAAV9/JeT-GAN therapy to refine the treatment. Next, investigators plan to test whether the GAN gene transfer is more effective when given to younger children or those in an earlier stage of the disease. The next phase of the trial will help to further determine its safety and efficacy.
More information: Diana X. Bharucha-Goebel et al, Intrathecal Gene Therapy for Giant Axonal Neuropathy, New England Journal of Medicine (2024). DOI: 10.1056/NEJMoa2307952