This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
New study explores next-generation vaccine technology for RSV
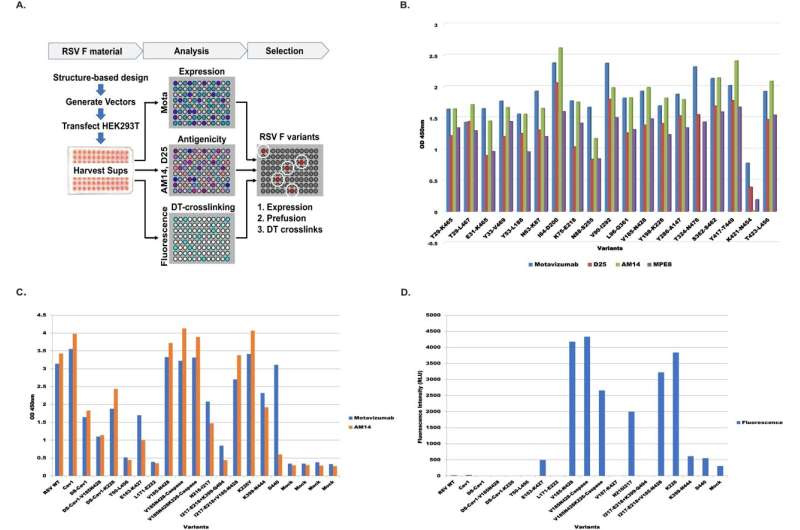
Calder Biosciences, Inc., a next-generation vaccine company, has published an article that debuts and validates the application of Calder's '3D Vaxlock' platform technology. When applied to the Respiratory Syncytial Virus (RSV) F protein as a vaccine immunogen, Calder's 3D Vaxlock technology achieves an unprecedented 11X more potent immune response than the standard industry comparator.
Application of the technology not only dramatically improves potency but also improves the shelf-life of the vaccine at refrigerated temperatures, which will facilitate distribution and delivery.
The 11-fold higher responses are measured in terms of antibodies generated that neutralize the virus on contact, thus preventing infection. Calder's vaccine also demonstrates improvements in the quality of immune responses elicited since a greater proportion of the antibodies produced neutralize the virus.
2023 was an exciting year for developments in preventing RSV, given the groundbreaking achievements of GSK and Pfizer in gaining licensure of the first-ever RSV vaccines, Arexvy and Abrysvo, for use in the elderly and maternal vaccination. While these licensures have already done a great deal to bolster human health, the published data suggest that adults over 75 represent a patient population that could benefit greatly from additional potency improvements.
"There remains an urgent need for vaccines that provide 75+ older adults and the frail good protection. And protecting newborn children for a longer period through maternal vaccination also remains an important goal," said Florian Schödel, MD, a member of Calder's Scientific Advisory Board. In the Nature Communications manuscript, Calder demonstrates that a high dose of their RSV vaccine achieves responses in aged, immunosenescent animals equivalent to responses in the young animal control group.
Calder's 3D Vaxlock technology engineers zero-length carbon-carbon bonds between tyrosine residues in target vaccine immunogens. This technology combines structure-based design and a novel bioprocess, and the bonds are site-specific and targeted to preserve the 3D shape of a protein in the regions that elicit the best immune responses, achieving best-in-class protection.
"A new stabilization technology as clean and precise as this one is exactly what's needed to unlock the full potential of protein vaccines," Calder's co-founder and CEO Chris Marshall stated to describe the potential of the technology. Unlike disulfide bonds, dityrosine bonds are irreversible, and their formation is catalyzed after the protein is fully folded in its native state.
In addition to the RSV program, Calder is applying its technology to Universal Influenza and Epstein-Barr virus vaccines. "On the heels of the COVID pandemic, the benefits of vaccination for human health are incontrovertible. Our technology is going to revolutionize the vaccine space, making vaccines that are even safer and more potent," said Calder's co-founder and CSO, Mark Yondola, who is also the senior author of the paper.
Calder also plans to apply its technology more generally in protein engineering, imparting thermostability as needed to additional subunit vaccines, biologics, or other targets.
The work is published in the journal Nature Communications.
More information: Sonal V. Gidwani et al, Engineered dityrosine-bonding of the RSV prefusion F protein imparts stability and potency advantages, Nature Communications (2024). DOI: 10.1038/s41467-024-46295-8