This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Hepatitis C virus infection: Research team discovers that a protein has an unexpected proviral effect
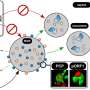
A research team from the Paul-Ehrlich-Institut has gained important insights into the role of the human guanylate-binding protein 1 (GBP1) in hepatitis C virus infection. Their results show that GBP1 performs a previously unknown function, that of a proviral factor, during the HCV life cycle.
Contrary to previous assumptions, the protein does not inhibit infection, but promotes it. GBP1 is induced by treatment with interferons. Therefore this research, published in PLOS Pathogens, indicates that interferon-based therapies should take the different modes of action of immunoregulatory factors into account.
The hepatitis C virus (HCV) is an RNA virus from the Flaviviridae family that can cause chronic liver disease, affecting approximately 58 million people worldwide. There are no vaccines against HCV available.
One result of HCV infection is that the immune system produces type I and III interferons (IFNs). The interferons subsequently mediate antiviral and immunoregulatory activities. IFNs induce certain molecules, such as guanylate-binding proteins (GBPs), which also include GBP1. There are a variety of reports describing the antiviral effect of IFN-inducible GBPs on various viruses.
Researchers, led by Dr. Daniela Bender in the working group of Professor Eberhard Hildt, Head of the Virology Division at the Paul-Ehrlich-Institut, dealt with the role of GBP1 in HCV infection in their latest work.
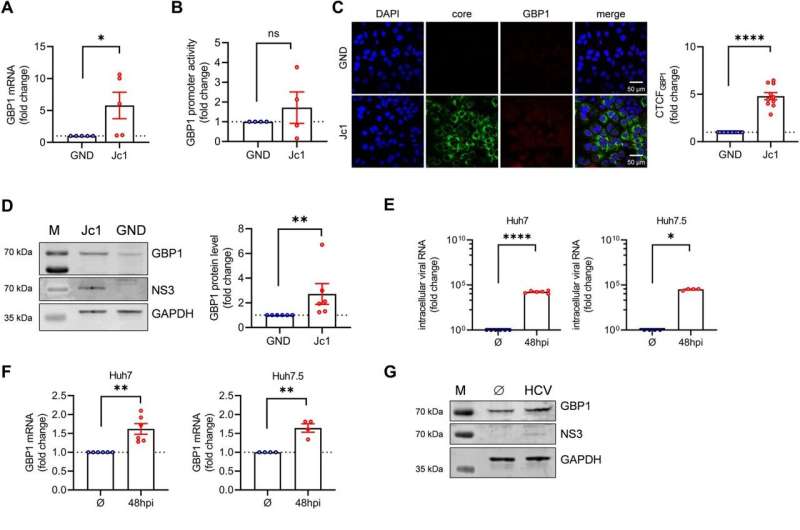
The researchers conducted a number of experiments in their studies, including real-time quantitative PCR (polymerase chain reaction), Western blot analyses, confocal laser scanning microscopy (CLSM), high-resolution STED (Stimulated Emission Depletion) microscopy, as well as virus titration and reporter gene assays. The team observed that HCV induces the expression—the reading of genes—of GBP1.
In addition, the research team found that—contrary to previous assumptions—the increased formation of GBP1 favors viral replication and release of HCV. Using CLSM and STED analyses, the researchers discovered that GBP1, together with the viral structural protein HCV core, is found near the nucleus in a network of membranes and lipid droplets.
The whole structure is part of a membrane network, which is a typical feature of HCV-infected cells. GBP1, along with HCV core, is found on the surface of the lipid droplets, where it binds factors involved in the formation of HCV particles. As a result, GBP1 promotes viral assembly and release of the hepatitis C viruses.
These research findings shed new light on the understanding of the interaction between HCV and the human host factor GBP1. They could potentially lead to new therapeutic approaches for the treatment of HCV infections that specifically intervene in the interactions between GBP1 and the virus. The findings should particularly be taken into account in the application of interferon-based therapies.
More information: Daniela Bender et al, Guanylate-binding protein 1 acts as a pro-viral factor for the life cycle of hepatitis C virus, PLOS Pathogens (2024). DOI: 10.1371/journal.ppat.1011976