April 8, 2024 dialog
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
written by researcher(s)
proofread
Brainstem neuronal shutdown as a potential cause of sudden death in at-risk infants
Sudden unexpected death in children, which includes sudden infant death syndrome (SIDS), is a devastating but poorly understood global health problem. The cause of SIDS was long considered a mystery, and although this mystery has never been solved, it was considered to be a sleep accident.
This is reflected by how the problem of SIDS has been addressed: Campaigns have successfully raised awareness among the public, most notably with the "Back to Sleep" campaign in the 1990s. As such, SIDS is often considered a suffocation event. However, data from epidemiological and genetic studies show that this is at best an incomplete explanation, indicating a clear need for research on the biological/medical mechanisms of SIDS and other sudden death syndromes.
To determine such a potential cause, we have studied a particular clinical case combined with laboratory results in a collaborative effort involving research teams from the Netherlands, the U.K. and France. The case concerned an infant presenting with episodes of sudden and severe apnea. These episodes were in multiple instances life-threatening and required resuscitation and hospital admissions. Genetic analyses revealed a mutation in the SCN1A gene. This gene has previously been associated with sudden death syndromes including SIDS.
Elucidating the mechanism for apnea in the laboratory
Since we presumed that the episodes of severe apnea could be caused by the SCN1A mutation, we performed laboratory experiments in a mouse model in which the same genetic mutation was introduced. The animals showed events of sudden and severe apnea similar to the clinical case, which were invariably fatal when we did not intervene. However, the animals recovered with the initiation of timely mechanical ventilation after the apnea.
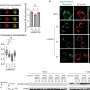
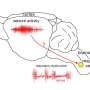
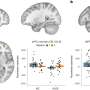
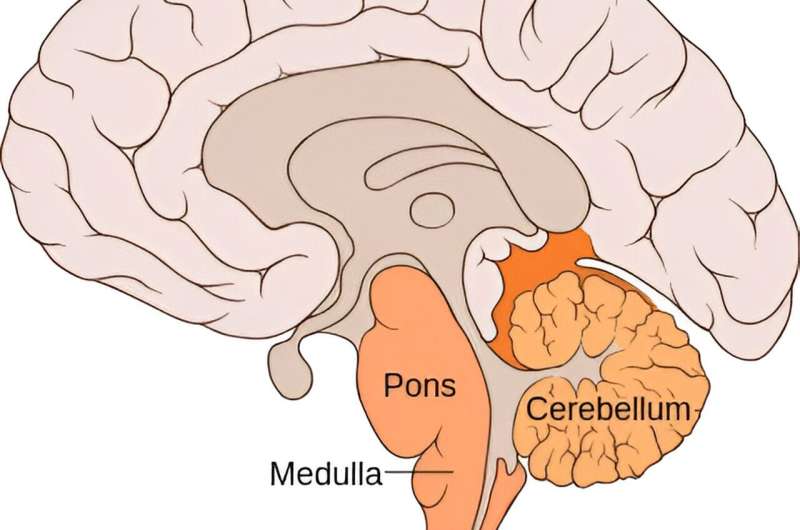
To investigate the link between apnea and the genetic mutation, we introduced the SCN1A mutation in cells. The mutation was found to increase the firing of these cells, indicating hyperexcitability. In the animals, we found that at the time of the sudden apnea, similar hyperexcitable conditions occurred in the brainstem.
These conditions lead to a sudden wave of neuronal firing that was followed by neuronal silence. Since brainstem areas that are necessary for generating the breathing rhythm were also involved, we were able to attribute the sudden apnea to neuronal shutdown in these areas. Our research is published in the Proceedings of the National Academy of Sciences.
Preventing apnea by preventing brainstem neuronal shutdown
Importantly, the cell studies also showed that sodium channel blockers, including carbamazepine, decreased the observed hyperexcitability. In the animals, these drugs prevented the sudden waves of neuronal firing and neuronal shutdown in the brainstem, which prevented apnea and death. Similarly, upon administering carbamazepine to the infant, the severity of apneic episodes decreased, obviating the need for resuscitation.
By linking the SIDS-associated SCN1A gene to events of sudden (lethal) apnea, we believe we have provided a research path in the search for causes of SIDS and other sudden death syndromes. In addition, the study shows that by preventing sudden brainstem neuronal shutdown, at-risk infants with similar genetic mutations may benefit from carbamazepine or other sodium channel blockers.
This story is part of Science X Dialog, where researchers can report findings from their published research articles. Visit this page for information about Science X Dialog and how to participate.
More information: Nico A. Jansen et al, Brainstem depolarization–induced lethal apnea associated with gain-of-function SCN1A L263V is prevented by sodium channel blockade, Proceedings of the National Academy of Sciences (2024). DOI: 10.1073/pnas.2309000121