This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers present new findings on the development of the human forebrain
A group of researchers at University of California San Diego School of Medicine led an investigation that offers new insight into the development of the human forebrain.
The study, led by Changuk Chung, Ph.D., and Xiaoxu Yang, Ph.D., both from the laboratory of Joseph G. Gleeson, M.D., at the School of Medicine Department of Neurosciences and the Rady Children's Institute for Genomic Medicine, provide a greater understanding of how the human brain develops at the cellular level.
The study also presents evidence for the existence of the source of inhibitory neurons (dInNs) in the human brain that differ from origins in other species like mice, a common lab animal used in brain studies. The group outlined their findings in a paper published in the journal Nature.
The forebrain, or cerebral cortex, is the largest part of the brain, important for a wide range of function, ranging from cognitive thought, vision, attention and memory. Neurons are cells that serve as the individual circuits of the brain. Inhibitory neurons usually function as a kind of neural "off" switch, as opposed to the "on" switch of excitatory neurons.
"Humans have a very large and wrinkled cortex that likely supports higher cognitive functions compared with other species, such as rodents," Gleeson explained.
He said that the inhibitory neurons in mice have an origin from deep within the developing brain. The current study puts that model to test by assessing cellular lineage. They found existence of dInNs, which are absent in mice. He said finding evidence for this specific type of neuron in humans opens the door to greater understanding how the human brain is special.
"We expect dInNs to support new, more accurate, models of human brains," Gleeson said. "This updated brain model may help explain the origins of certain conditions like epilepsy, schizophrenia or autism."
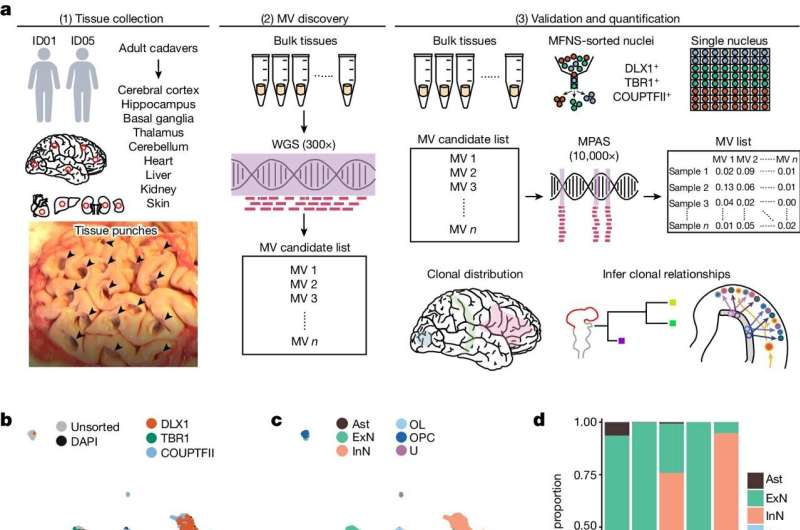
The group was especially interested in following the lineage trail of mosaic variants of brain cells. "If two cells share the same mother cell, we say they have the same lineage," Chung said.
"If two individual cells have a same mosaic variant, they were born from a common mother cell that passed it to all of its daughters," Yang explained. "So, mosaic variants in cells function like family names in people."
The researchers directly accessed brains from two neurotypical donors who died from natural causes. They used mosaic variants to trace where these cells came from, to identify sister cells born in the same brain region, and to determine how far each "family name" spread across the brain.
They revealed that some inhibitory and excitatory neurons essentially have the same family name, which Chung said means the two types of neurons share lineage. The two types likely branched in a late moment of embryonic cerebral development, he added, noting that such a cellular relationship is not present in other species.
"We hope our paper helps other researchers generate better models of neurological disease, and which types of brain diseases can result from impaired development," Gleeson concluded.
More information: Changuk Chung et al, Cell-type-resolved mosaicism reveals clonal dynamics of the human forebrain, Nature (2024). DOI: 10.1038/s41586-024-07292-5