This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Mathematical model exploits cancer cell plasticity to guide adaptive therapy
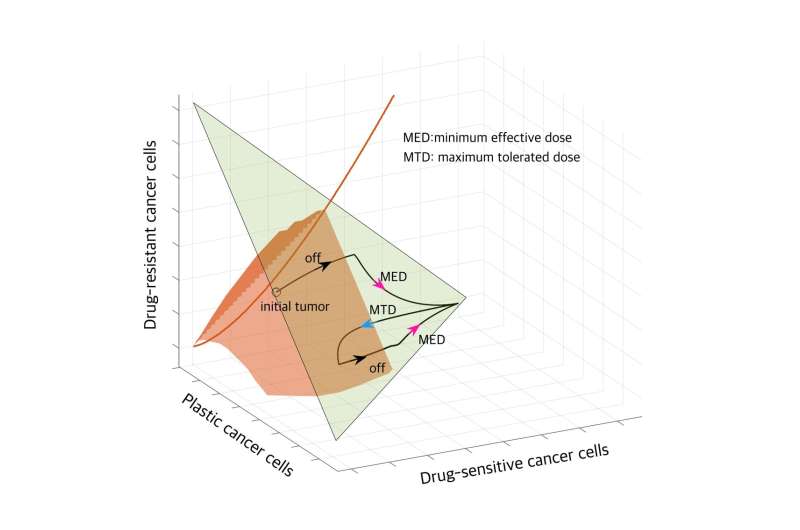
Cancer poses significant challenges due to the development of resistance and the likelihood of relapse. Resistance may arise from permanent genetic changes in cancer cells or non-genetic alterations in cancer cell behavior induced by treatment. Standard of care in cancer treatments typically involves administering the maximum tolerated dose of a drug to eradicate drug-sensitive cells effectively. However, this approach often fails in the long term because drug-resistant cancer cells can grow more rapidly when all drug-sensitive cancer cells are killed off.
An evolution-based treatment approach, called adaptive therapy, personalizes treatment dose or breaks based on individual patient responses. The goal of adaptive therapy is to maintain a sufficient number of sensitive cells to control the growth of resistant cells. Recent studies and clinical trials have demonstrated that adaptive therapy could delay resistance more effectively compared to the standard of care.
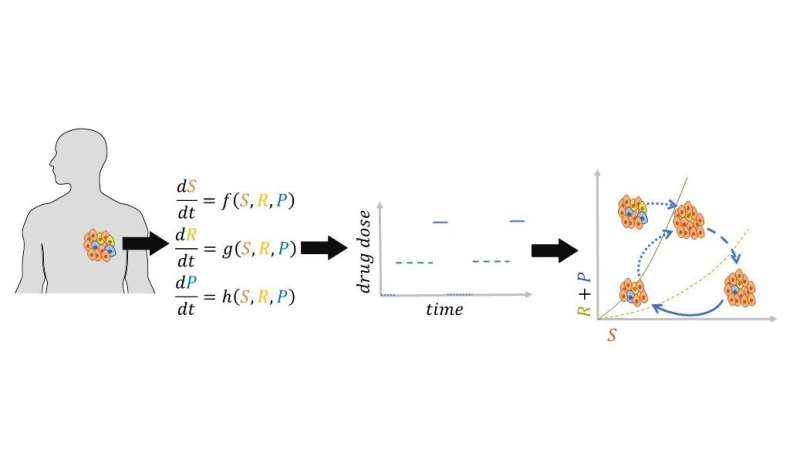
Determining the dose and treatment breaks for each patient is challenging because cancer is a complex evolving system, and every patient is different. Mathematical models can be helpful in designing such patient-specific treatment strategies. Indeed, several mathematical models have been developed to explore the effects of various treatment strategies on patient outcomes.
However, existing mathematical models often overlook the impact of acquired resistance and cancer cell plasticity. "Acquired resistance" encompasses various types of resistance that emerge, often due to genetic changes. "Cell plasticity" refers to cancer cells' ability to alter their phenotypes in response to changes in their microenvironment, such as fluctuations in treatment dosage or treatment cessation
A research team led by Dr. Kim Eunjung at the Natural Products Informatics Research Center of the Korea Institute of Science and Technology (KIST) has established a theoretical foundation for cancer treatment strategies following tumor evolution. The work is published in the journal Chaos, Solitons & Fractals.
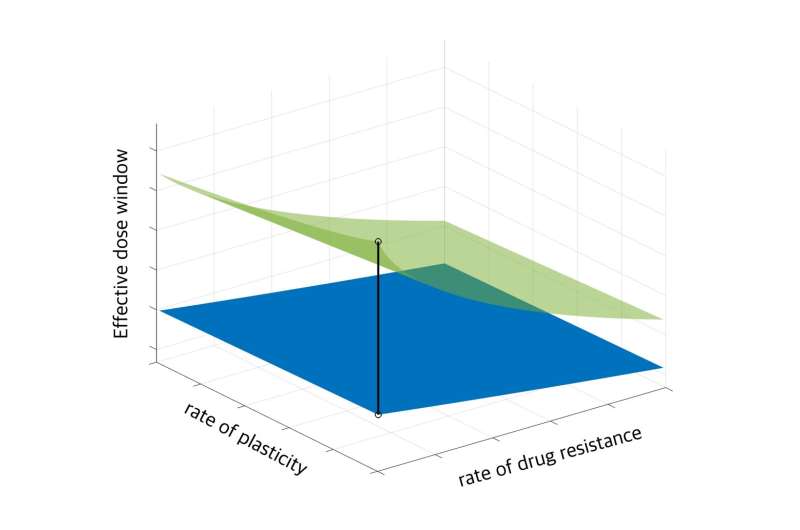
They have developed a mathematical model to predict tumor evolution, considering the acquisition of resistance by cancer cells and their ability to alter phenotypic behavior (plasticity) during treatment. The analysis of their model has identified the conditions for the existence of an effective dose window, a range of doses that could maintain tumor volume at an equilibrium point, where the tumor volume remains unchanged and stable.
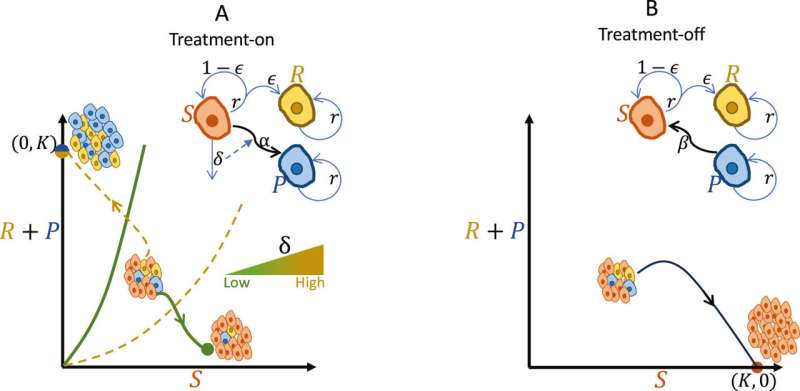
For some tumors with plasticity, taking breaks from treatment helps cancer cells become sensitive again, joining forces with other sensitive cells to suppress resistant cell growth. The research team has proposed evolutionary therapy dosing, which involves administering treatment in cycles comprising treatment holidays, minimum effective doses, and maximum tolerated doses.
Pausing treatment allows plastic cancer cells to regain sensitivity, followed by the application of a minimum effective dose to control tumor volume. Subsequently, a maximum tolerated dose is administered to further reduce tumor size. This dosing cycle effectively contains tumor volume at a manageable level.
Numerical simulations of the proposed strategies, applied to a melanoma patient, further illustrate these findings. The results show that evolutionary dosing can redirect tumor dynamics, maintaining tumor size below a tolerable burden.
The developed mathematical model can predict the effective dosage range of cancer treatment drug candidates prior to clinical trials. It can assist in determining the anticancer effects of new treatments and identifying the effective dosage range for each drug. Furthermore, the model contributes to personalized cancer treatment strategies by considering patient-specific tumor evolutionary dynamics during treatment.
Dr. Kim Eunjung stated, "In the current study, we emphasized the role of cancer cells' phenotypic plasticity in enhancing the controllability of tumor burden with evolutionary treatment cycling doses."
She also mentioned plans to utilize the mathematical model in designing animal experiments and clinical trials for potential natural product-derived anticancer drug candidates. This aims to establish dosage regimens that effectively control tumor burden.
More information: M.A. Masud et al, Theoretical understanding of evolutionary dosing following tumor dynamics, Chaos, Solitons & Fractals (2024). DOI: 10.1016/j.chaos.2024.114451

















