This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
reputable news agency
proofread
Study finds 1.5 percent ruxolitinib cream safe, effective for teens with eczema

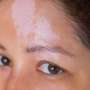
Long-term intermittent use of ruxolitinib cream is well tolerated and provides disease control in adolescent patients with atopic dermatitis (AD), according to a study published online May 2 in the American Journal of Clinical Dermatology.
Lawrence F. Eichenfield, M.D., from the University of California San Diego, and colleagues assessed the safety and efficacy of 1.5% ruxolitinib cream versus vehicle and long-term disease control of ruxolitinib cream among adolescents using pooled data from phase 3 studies. The analysis included 245 adolescents (aged 12 to 17 years) with AD for at least two years.
The researchers found that at week 8, substantially more patients who applied 1.5% ruxolitinib cream versus vehicle achieved Investigator's Global Assessment (IGA) treatment success (50.6 versus 14.0%), ≥75% improvement in the Eczema Area and Severity Index (60.9 versus 34.9%), and a 4-point improvement or greater in the itch numerical rating scale (52.1 versus 17.4%).
During the long-term safety period, mean trough steady-state ruxolitinib plasma concentrations at weeks 12 and 52 were 27.2 and 15.5 nM, respectively. With ruxolitinib, the percentage of patients achieving an IGA score of 0 or 1 was sustained or further increased. Application site reactions through 52 weeks occurred in 1.8% of participants applying 1.5% ruxolitinib cream at any time. No patients reported serious adverse events.
"Treatment with 1.5% ruxolitinib cream in adolescent patients with mild-to-moderate atopic dermatitis had anti-inflammatory and antipruritic effects comparable with those observed in the overall study population," the authors write.
The study was funded by Incyte, the manufacturer of ruxolitinib.
More information: Lawrence F. Eichenfield et al, Efficacy, Safety, and Long-Term Disease Control of Ruxolitinib Cream Among Adolescents with Atopic Dermatitis: Pooled Results from Two Randomized Phase 3 Studies, American Journal of Clinical Dermatology (2024). DOI: 10.1007/s40257-024-00855-2
© 2024 HealthDay. All rights reserved.