This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
reputable news agency
proofread
Study shows crinecerfont aids patients with congenital adrenal hyperplasia
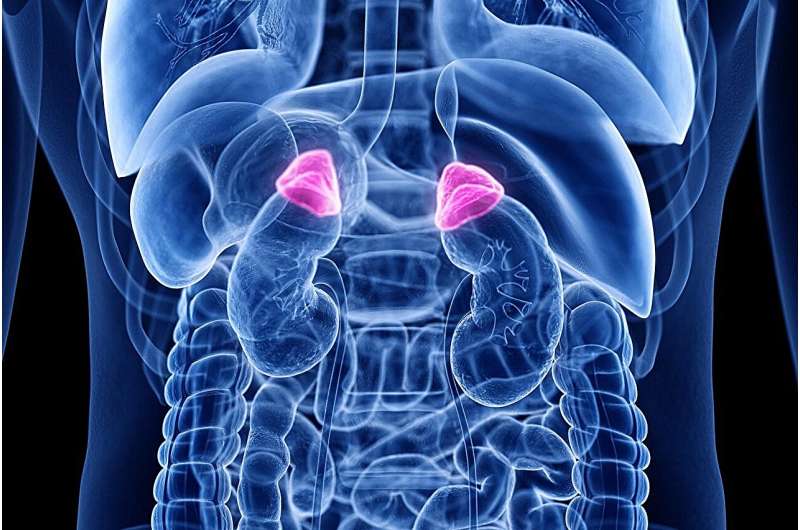
Crinecerfont results in a greater decrease from baseline in the mean daily glucocorticoid dose among patients with congenital adrenal hyperplasia (CAH), according to a study published June 2 in the New England Journal of Medicine to coincide with the annual meeting of the Endocrine Society, held from June 1 to 4 in Boston.
Richard J. Auchus, M.D., Ph.D., from the University of Michigan Medical School in Ann Arbor, and colleagues randomly assigned adults with CAH to receive crinecerfont or placebo for 24 weeks in a 2:1 ratio (122 and 60 patients, respectively).
To assess androstenedione values, glucocorticoid treatment was maintained at a stable level for four weeks, followed by glucocorticoid dose reduction and optimization over 20 weeks to achieve the lowest glucocorticoid dose that maintained androstenedione control.
The researchers found that at week 24, the change in the glucocorticoid dose was −27.3 and −10.3% in the crinecerfont and placebo groups, respectively (least-squares mean difference, −17.0%age points). Overall, 63 and 18% of patients in the crinecerfont and placebo groups, respectively, reported a physiologic glucocorticoid dose.
At week 4, there was a decrease observed in androstenedione levels with crinecerfont and an increase with placebo (−299 and 45.5 ng/dL, respectively; least-squares mean difference, −345 ng/dL). The most common adverse events in the two trial groups were fatigue and headache.
"Crinecerfont therapy allowed for a substantial and clinically meaningful reduction in glucocorticoid administration to more physiologic doses in adults with classic CAH," the authors write.
Several authors disclosed ties to biopharmaceutical companies, including Neurocrine Biosciences, which manufactures crinecerfont and funded the study.
More information: Richard J. Auchus et al, Phase 3 Trial of Crinecerfont in Adult Congenital Adrenal Hyperplasia, New England Journal of Medicine (2024). DOI: 10.1056/NEJMoa2404656
© 2024 HealthDay. All rights reserved.